Q. What is conversion of ammonia into nitrates called? Ans: Nitrification Q. State the role of the atmosphere in climate control? Ans: Atmosphere prevents sudden increase in temperature during the day light hours. It slows down the escape of heat into outer space during the night. Q . How following factors contribute in formation of soil ? (a) wind (b) water (c) Sun Ans: Wind : Strong winds erode rocks down. The strong wind carries small rock pieces and sand from one place to other like water. Water : (1) Fast flowing water carries big and small particles of rock downstream. These rocks rub against other rocks and the resultant abrasion causes rocks to wear into smaller and smaller particles (2) Water expands and freezing. So when it is deposited in crevices of rocks it cracks rocks into smaller pieces. Sun : The sun heats up rocks during the day so they expand. At night these rocks cool down and contract. This results in the formation of cracks and ultimately the huge rock breaks into smaller pieces. Q. Acid rain and smog are said to be the consequences of air pollution. How are they caused? What are the ill effects of breathing polluted air on human health? Ans: Oxides of nitrogen and sulphur (air pollutants) dissolve in rain and form HNO3 and H2SO4 making the rain acidic Suspended unburnt carbon particles (hydrocarbon) cause visibility to be lowered when water condenses during cold weather – Smog. Ill effects – allergies, respiratory problems cancer, heart disease Q. What is Smog? Ans: Presence of suspended particles like carbon particles or hydrocarbons causes visibility to be lowered, especially in cold weather, when water also condenses out of air. This is called smog. Q. What is green house effect? List two green house gases. State the ultimate effect of increase in green house gases in the environment. Ans: The warming up of the atmosphere due to trapped radiation is called green house effect. Green house gases carbon dioxide methane increase in green house gases will lead to global warming Q. What makes the biosphere dynamic but stable system ? Ans: A constant interaction between biotic and abiotic components of biosphere makes it dynamic and stable. Interaction consists of transfer of matter and energy between different components of biosphere. Q. The atmosphere acts as a blanket. How ? Ans: Air in the atmosphere is a bad conductor of heat and it keeps the average temperature of the earth fairly steady during the day and the whole year. Q. What is soil? How is it formed? State the major factor that decides the structure of a soil. What role does it play ? Ans: Soil: It is a mixture containing small particles rocks and lots of decayed living organism called human. Formation of soil: Weathering of rocks Major factor – Humus. It makes the soil more process that water penetrates deep underground. Q. Write the importance of ozone in the atmosphere. Ans: Ozone layer is present in the stratosphere of the atmosphere. This layer prevents harmful radiations like ultraviolet radiation from reaching the surface of the earth where they may damage many forms of life. Q. (a) List two activities of man which lead to environmental pollution. (b) List any two uses of carbon in living organisms. Ans: (a)Release of harmful gases from vehicles. Excessive use of chemicals in agriculture. Excessive combustion of fossil fuels. (b) Carbon dioxide is used by plants during photosynthesis to synthesise food. Carbon is present in molecules like protein, carbohydrates and fats needed by organisms for its biochemical cycle. Q. List two forms of oxygen found in the atmosphere. Name the process(s) by which (i) oxygen from the atmosphere is used up. (ii) oxygen is returned to the atmosphere Ans: Diatomic oxygen (O2) and tri atomic oxygen (O3) Oxygen is used up by the processes : combustion, respiration and in the formation of oxides of nitrogen. Oxygen is returned to the atmosphere by photosynthesis. Q. State various steps and processes involved in the nitrogen cycle in nature. Also show cycling of various nutrients in this cycle. Ans: (1) Nitrogen fixation Conversion of atmospheric nitrogen into nitrates and nitrites by Rhizobium 2) Ammonification : Death and decay of plant bodies releases ammonia. Animals give out ammonia, urea etc. These nitrogenous compounds which are converted to ammonia. (3) Nitrification : Ammonia converted into nitrites and then nitrates by nitrifying bacteria. (4) Denitrification : Pseudomonas convert nitrates and nitrites into elemental nitrogen. Q. Write a note on how forests influence the quality of air, soil and water resources. Ans: Air: (1) Minimising level of CO2. (2) Reduces temperature (3) Trees ability to absorb harmful gases. Soil : (1) Prevent soil erosion (2) Regulate Biogeochemical cycles. (3) Influence rain hence increase ground water level. Q. How is atmosphere on our Earth different from the atmosphere on Venus and Mars ? State two ways by which percentage of carbon dioxide is fixed on the earth. Ans: On the planets Venus and Mars carbon dioxide forms the major component constituting up to 95-97% of the atmosphere. On the contrary the major component of the earth’s atmosphere is air which consists of nitrogen, oxygen, carbon dioxide, argon, water vapour and trace components. Moreover, unlike Venus and Mars earth has life on it. Two ways by which percentage of CO2 is fixed on earth are : (i) Green plants convert CO2 into glucose in the presence of sunlight (ii) Many marine animals use carbonates dissolved in sea water to make their shells. Q. “Urbanization and industrialisation is mainly responsible for the increase in environment pollution” Justify this statement and suggest ways and means to check it. Ans: (a) 1. Increased the consumption of fossil fuel – increased the production of pollution like CO, SO2, NO2, CO2 etc. 2. CFC has led to deplection in ozone layer results in one entry of U.V rays in to earth’s atmosphere. 3. Acid rain 4. Production of green house gases (b) Measures : 1. Reduce consumption of fossil fuels 2. Planting more trees 3. Using Isolentanes instead of CFC 4. Usage of non conventional sources of energy e.g solar, wind, tidal energy Download CBSE Summative & Formative Assignment based on CCE Biology Term-2 Biology Science Ch-07. Diversity in Living Organisms Download File Biology Science Ch-13. Why do we fall ill Download File Biology Science Ch-14. Natural Resources Download File Download CBSE Summative & Formative Assignment based on CCE Test Paper Biology Term-2 CCE Test Paper Biology Chapter Natural Resources ix Biology-1 Download File CCE Test Paper Biology Chapter Natural Resources ix Biology-2 Download File Class IX Biology Chapter 7 : Diversity In Living Organisms NCERT Solutions Download File CCE Test Paper Biology Chapter 7 : Diversity In Living Organisms Solved Download File 9th_diversity_in_living_organization_study_notes. Download File Class IX Biology Chapter Chapter: Why do we fall ill? NCERT Solutions Download File CCE Test Paper Biology Chapter: Why do we fall ill? Solved Download File
10 Comments
 After completing this lesson, you will be able to:
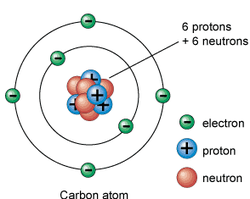
The word atom originally meant a smallest possible particle of matter, not further divisible. Democritus 1. The existence of atoms was proposed as early as in the 5th century BC by the Greek philosophers Leucippus and his pupil Democritus, for which they were called atomists. 2. Democritus, develop the idea of atoms. He asked this question: If you break a piece of matter in half, and then break it in half again, how many breaks will you have to make before you can break it no further? . Democritus thought that it ended at some point, a smallest possible bit of matter. He called these basic matter particles, atoms. 4.The word "atom" is derived from the Greek word "atomos", which means indivisible". John Dalton 1. Five main points of Dalton's Atomic Theory • All matter is composed of extremely small particles called atoms. • All atoms of a given element are identical, having the same size, mass, and chemical properties. Atoms of a specific element are different from those of any other element. • Atoms cannot be created, divided into smaller particles, or destroyed. • Different atoms combine in simple whole-number ratios to form compounds. • In a chemical reaction, atoms are separated, combined, or rearranged. Weakness 1. Atoms consist of even smaller particles called electrons, protons and neutrons. 2. Atoms can be created and destroyed in the nuclear reactions such as nuclear fusion and nuclear fission. Atoms of the same element can have different physical properties, for example, isotopes of hydrogen. J.J. Thomson Plum pudding model 1. In physics, the Plum pudding model of the atom was made after the discovery of the electron and was proposed by the discoverer of the electron, J. J. Thomson. 2. In it, the atom is envisioned as electrons surrounded by a soup of positive charge, like plums surrounded by pudding. 3. The electrons were positioned uniformly throughout the atom. 4. Instead of a soup, the model is also said to have had a cloud of positive charge. 5. This model can be compared to a British treat called plum pudding, hence the name. It is also known as the chocolate chip cookie model. Ernest Rutherford Gold foil experiment The Gold foil experiment, or Geiger-Marsden experiment was an experiment done by Hans Geiger and Ernest Marsden in 1909, under the direction of Ernest Rutherford at the Physical Laboratories of the University of Manchester which led to the downfall of the plum pudding modelof the atom. 1. They measured the deflection of alpha particles directed normally onto a sheet of very thin gold foil. 2. Under the prevailing plum pudding model, the alpha particles should all have been deflected by at most of a few degrees. 3. However they observed that a very small percentage of particles were deflected through angles much larger than 90 degrees. 4. From this Rutherford concluded that the atom contained a very small positive charge which could repel the alpha particles if they came close enough. Rutherford Atom 1. Early in 1911 Rutherford published a revised model of the atom, known as the Rutherford atom. 2. He concluded that • the atom is mostly empty space, • most of the atom's mass concentrated in a tiny center, the nucleus and electrons being held in orbit around it by electrostatic attraction. • The nucleus was around 10-15 meters in diameter, in the centre of a 10-10 metre diameter atom. • Those alpha particles that had come into close proximity with the nucleus had been strongly Model: Solar System Model (Bohr’s Model) 1. Niels Bohr improved on Rutherford's atomic model. 2. Bohr model depicts the atom as a small, positively charged nucleus surrounded by electrons in orbit - similar in structure to the solar system, but with electrostatic forces providing attraction, rather than gravity. 3. According to Bohr’s Model • Electrons in an atom of an element are not randomly distributed around the atomic nucleus. • Electrons move around the nucleus in fixed orbits.• Each orbit forms a circle and has a fixed distance from the nucleus. James Chadwick’s Model In 1932, James Chadwick bombarded beryllium (Be) with alpha aprticles. He allowed the radiation emitted by beryllium to incident on a paraffin wax. It was found that protons were shot out form the paraffin wax. People began to look for what was in the "beryllium radiations". 1. Chadwick discovered the presence of neutrons in the nucleus. 2. He concluded that the nucleus contains another tiny particle known as a neutron that has nocharge. 3. The neutron mass is almost similar to the proton mass. 4. All nuclei contain protons and neutrons, except for the hydrogen which contains protons. only Modern Atomic Model 1. The atomic model in the present day is based on the contributions of the above scientists. 2. According to the modern atomic model, (a) The central nucleus consists of protons and neutrons. It containing almost all the mass of the atom. (b) the nucleus of an atom is very small compared to the size of the atom (c) the electrons are orbiting outside the nucleus in the electron shells(d) the electrons are moving in electron shells at a very high speed and we cannot determine the position of the electrons at a particular time The subatomic particles of an atom 1. Atoms are made up of tiny particles called subatomic particles. 2. An atom contains three types of subatomic particles:a. proton,b. neutron andc. electron, 3. The proton and neutron form the nucleus at the centre of an atom. 4. The electron moves around the nucleus at a very high speed. 5. The nucleus is positively charged because of the presence of protons, which are positively charged. The neutrons are neutral. Proton number and nucleon number Proton Number 1. The proton number (Z) represent the number of protons found in the nucleus of an atom. Proton number = the number of protons 2. The proton number is also known as the atomic number. 3. In an atom of neutral charge, the number of electrons also equals the atomic number. 4. Hence, the proton number of an atom can also represent the number of electrons. Nucleon Number 1. The nucleon number (A), also called atomic mass number or mass number, is the number ofprotons plus the number of neutrons in an atomic nucleus. Nucleon number = Number of protons + Number of Neutrons The nucleon number of an atom is about the same as the mass of the atom because the mass of anelectron is very small and can be ignored. Continued>>>>>>>>>> Read more to excel IX Atomic Structure ( a fundamental particles) Discovery of Electron 9th Atomic Structure (Discovery of a fundamental particle protons) 9th Atoms and Molecules 9th Structure of Atoms Practice paper and sample paper based On Atomic structure Discovery of electrons, protons and neutrons Download File
Discovery of Electron Who discovered the electron? Electron was discovered by J. J. Thomson in 1897 when he was studying the properties of cathode ray. What is cathode ray? J. J. Thomson constructed a glass tube which was partially evacuated i.e. much of the air was pumped out of the tube. Then he applied a high electrical voltage between two electrodes at either end of the tube. He detected that a stream of particle (ray) was coming out from the negatively charged electrode (cathode) to positively charged electrode (anode). This ray is called cathode ray and the whole construction is called cathode ray tube. The schematic of a cathode ray tube Conclusion s: o Cathode rays consist of electrons. o Electrons are the basic unit s of all atoms. Properties of cathode ray particle 1. They travel in straight lines. 2. They are independent of the material composition of the cathode. 3. Applying electric field in the path of cathode ray deflects the ray towards positively charged plate. Hence cathode ray consists of negatively charged particles. J. J. Thomson measured the charge-by-mass-ratio (e/m) of cathode ray particle using deflection in both electric and magnetic field. e/m =−1.76×108 coulomb per gram The cathode ray particle turned out to be 2000 times lighter than hydrogen. Although we got e/m ratio for electron from J.J. Thomson’s Cathode Ray Tube experiment, we still don’t know the exact charge (e) for electron. American physicist Robert Millikan designed an experiment to measure the absolute value of the charge of electron which is discussed below. Millikan Oil Drop Experiment In 1909, American physicist R. Millikan measured the charge of an electron using negatively charged oil droplets. The measured charge (e) of an electron is −1.60×10−19 Coulombs. Using the measured charge of electron, we can calculate the mass of electron from e/m ratio given by J. J. Thomson’s cathode ray experiment. e/m=−1.76×108 Coulomb-per-gram m=e−1.76×108 Putting e=−1.60×10−19 Coulomb, m=9.1×10−28 gram. What we have learned Electron was discovered by J. J. Thomson in Cathode Ray Tube (CRT) experiment. Electrons are negatively charged particles with charge-to-mass ratio −1.76×108 C/gm The charge of an electron was measured by R. Millikan in Oil drop experiment. Charge of an electron is −1.60×10−19 C Mass of an electron is 9.1×10−28 gram. Electron is approximately 2000 times lighter than hydrogen. Discovery of Proton
Atom: It is the smallest particle of an element which may or may not have independent existence. The atoms of certain elements such as hydrogen, oxygen, nitrogen, etc .do not have independent existence whereas atoms of helium, neon, argon, etc. do have independent existence. Thus we can say that all elements are composed of atoms. IUPAC (International Union of Pure and Applied Chemistry) approves names of elements. Many of the symbols are the first one or two letters of the element’s name in English. The first letter of a symbol is always written as a capital letter (uppercase) and the second letter as a small letter (lowercase) For example : (i) hydrogen, H (ii) aluminium, Al and not AL (iii) cobalt, Co and not CO. Symbols of some elements are formed from the first letter of the name and a letter, appearing later in the name. Examples are: (i) chlorine, Cl, (ii) zinc, Zn etc. Other symbols have been taken from the names of elements in Latin, German or Greek. For example, the symbol of iron is Fe from its Latin name ferrum, sodium is Na from natrium, potassium is K from kalium. Therefore, each element has a name and a unique chemical symbol. Molecule: A molecule is the smallest or the simplest structural unit of an element (or) a compound which contains one (or) more atoms. It retains the characteristics of an element. A molecule can exist freely and it is a combined form of bonded units whereas an atom is a singular smallest form of non bonded unit. Molecules are of two types, namely homo atomic molecules and hetero atomic molecules. Homo atomic molecules: These are the molecules which are made up of atoms of the same element. For example hydrogen gas consists of two atoms of hydrogen (H2).Similarly oxygen gas consists of two atoms of oxygen (O2). HETERO ATOMIC MOLECULES : The hetero atomic molecules are made up of atoms of different elements. They are also classified as diatomic, triatomic, or polyatomic molecules depending upon the number of atoms present. H2O, NH3, CH4, etc., are the examples for hetero atomic molecules. Atomicity: The number of atoms present in one molecule of an element is called the atomicity of an element. Depending upon the number of atoms in one molecule of an element, molecules are classified into monoatomic, diatomic, triatomic or poly atomic molecules containing one, two, three, or more than three atoms respectively. Mon atomic molecules: Helium (He) Neon (Ne) Metals Di atomic molecules: Hydrogen H2 Chlorine Cl2 Tri atomic molecules: Ozone (O3) Poly atomic molecules: phosphorous P4 Sulphur S8 Atomicity = Molecular Mass/Atomic mass MORE TO KNOW : Isotopes ⇒ These are the atoms of same element with same atomic number (Z) but different mass number (A). Example (17Cl35,17Cl37 ) Isobars ⇒ These are the Atoms of the different element with same mass number but different atomic number. Example (18Ar40, 20Ca 40 ) Isotones ⇒ These are the atoms of different elements with same number of neutrons Example : (6C13, 7N14 ) AVOGADRO’S HYPOTHESIS : Amedeo Avogadro put forward hypothesis and is based on the relation between number of molecules and volume of gases that is “volume of a gas at a given temperature and pressure is proportional to the number of particles”. Avogadro’s Law: Equal volumes of all gases under the same conditions of temperature and pressure. contain the equal number of molecules. TEST YOUR UNDERSTANDING SKILL (a) Find the atomicity of chlorine if its atomic mass is 35.5 and its molecular mass is 71 (b) Find the atomicity of ozone if its atomic mass is 16 and its molecular mass is 48 WHAT IS AN ION? An ion is a charged particle and can be negatively or positively charged. A negatively charged ion is called an ‘anion’ and the positively charged ion, a ‘cation’. For example, sodium chloride (NaCl). Its constituent particles are positively charged sodium ions (Na+) and negatively charged chloride ions (Cl–). Ions may consist of a single charged atom or a group of atoms that have a net charge on them. A group of atoms carrying a charge is known as a polyatomic ion e.g. Calcium oxide (Ca+2 O-2) RELATIVE ATOMIC MASS (RAM): Relative atomic mass of an element is the ratio of mass of one atom of element to the 1/12th part of mass of one atom of carbon. Relative atomic mass is a pure ratio and has no unit. If the atomic mass of an element is expressed in grams, it is known as gram atomic mass. e.g., Gram atomic mass of hydrogen = 1g ; Gram atomic mass of carbon = 12g Gram atomic mass of nitrogen = 14g ; Gram atomic mass of oxygen = 16g Atomic mass is expressed in atomic mass unit (amu). One atomic mass unit is defi ned as 1/12th part of the mass of one atom of carbon. Chemical Formulae: The chemical formula is a symbolic representation of a compound of its composition. Valency : The combining power (or capacity) of an element is known as its valency. Valency can be used to find out how the atoms of an element will combine with the atom(s) of another element to form a chemical compound. RELATIVE MOLECULAR MASS (RMM) : The relative molecular mass of an element or a compound is the ratio of mass of one molecule of the element or a compound to the mass of 1/12 th part of mass of one atom of carbon. Relative Molecular mass is a pure ratio and has no unit. If the molecular mass of a given substance is expressed in gram, it is known as gram molecular mass of that substance. Molecular mass is the sum of the masses of all the atoms present in one molecule of the compound or an element. Test your numerical skill : Problem: Find the gram molecular mass of water (H2O) Solution: Þ 2(H) = 2 x 1 = 2 and 1(O) = 1 x 16 = 16 Gram molecular mass of H2O = 2 + 16 = 18g Problem: Find the gram molecular mass of carbon dioxide Solution: Þ (CO2) 1(C) = 1 x 12 = 12 and 2(O) = 2 x 16 = 32 Gram molecular mass of CO2 = 12 + 32 = 44 g MOLE CONCEPT While performing a reaction, to know the number. of atoms (or) molecules involved, the concept of mole was introduced. The quantity of a substance is expressed in terms of mole. Definition of mole : Mole is defined as the amount of substance that contains as many specifi ed elementary particles as the number of atoms in 12g of carbon-12 isotope. One mole is also defined as the amount of substance which contains Avogadro number (6.023 x 1023) of particles. MORE TO KNOW Avogadro number: Number of atoms or molecules or ions present in one mole of a substance is called Avogadro number. Its value is 6.023 x 1023. Therefore, one mole of any substance = 6.023 x 1023 particles may be atoms, molecules, ions For e g. One mole of oxygen atoms represents 6.023 x 1023 atoms of oxygen and 5 moles of oxygen atoms contain 5 x 6.023x1023 atoms of oxygen. To find the number of moles, the following formulae are useful 1. When the mass of the substance is given: Use this formula : Number of moles = given mass/ atomic mass (a). Calculate the number of moles in (i) 81g of aluminium ii) 4.6g sodium (iii) 5.1g of Ammonia (iv) 90g of water (v) 2g of NaOH Solution: (i) Number of moles of aluminium = given mass of aluminium / atomic mass of aluminium = 81/27 = 3 moles of aluminium [Rest Question do yourself] (b) Calculate the mass of 0.5 mole of iron Solution: mass = atomic mass x number of moles = 55.9 x 0.5 = 27.95 g Do yourself : Find the mass of 2.5 mole of oxygen atoms [ Mass = molecular mass x number of moles] 2. Calculation of number of particles when the mass of the substance is given: Number of particles =( Avogadro number x given mass)/gram molecular mass Problem: Calculate the number. of molecules in 11g of CO2 Solution: gram molecular mass of CO2 = 44g Number of molecules = (6.023 x 1023 x 11) / 44 = 1.51 x 1023 molecules Do yourself: Calculate the number of molecules in 360g of glucose 3. Calculation of mass when number of particles of a substance is given: Mass of a substance = (gram molecular mass x number of particles)/6.023 x 1023 Problem: Calculate the mass of 18.069 x 1023 molecules of SO2 Solution: Gram molecular mass SO2 = 64gm The mass of 18.069 x 1023 molecules of SO2 = (64x18.069 x 1023)/ (6.023 x 1023) = 192 g Do yourself: (a) Calculate the mass of glucose in 2 x 1024 molecules (b) Calculate the mass of 12.046 x 1023 molecules in CaO 4. Calculation of number of moles when you are given number of molecules: Problem: Calculate the number moles for a substance containing 3.0115 x 1023 molecules in it. Solution: Number of moles = [Number of molecules/(6.023 x 1023)] = ( 3.0115 x 1023)/( 3.0115 x 1023) =0.5 moles Do yourself: (a) Calculate number of moles in 12.046x 1022 atoms of copper (b) Calculate the number of moles in 24.092 x 1022 molecules of water. Problem: Calculate the number of aluminum ions present in 0.051 g of aluminum oxide. (Hint: The mass of an ion is the same as that of an atom of the same element. Atomic mass of Al=27 u) Solution: Mass of the 1 mole of Al2 O3 = 2x27 + 3x16 = 102gm The number of ions present in 102 gm of aluminum oxide = 6.023 x 1023 ion The number of ions present in 0.051g of aluminum oxide= (6.023 x 1023 ion x 0.051g)/ 102 gm = 6.023 x 1023 ion x0.0005 = 3.0115 x 1020 ions In Al2 O3, Aluminium and oxygen are in ratio 2:3 So, The number of aluminum ions present(Al3+) in 0.051g of aluminum oxide = 2 x 3.0115 x 1020 ions =6.023 x 1020 ion DOWNLOAD FULL E-NOTES
Related posts
Ø IX (9th)Chemistry Ch 03: Solved Questions paper Ø IX (9th)Chemistry Ch 03: Solved Questions Ø 9th Atoms and molecules Study Notes, Q & A Ø CBSE Test paper-1 Ø CBSE Test paper-2 Ø CBSE Test paper-3 Ø Numerical based on mole concept Ø Solved questions For FA-3 Ø Self-evaluation on Mole concept Ø Question bank By CBSE Pathshala For CBSE Ø Guess Questions Contents: 1. Thrust and Pressure 2. Pressure in Fluids 3. Buoyancy 4. Why objects sink or Float when placed on surface of water? 5. Archimedes Principle 6. Relative Density Learning Objectives: The students will: 1. Understand the difference between Thrust and Pressure. 2. Apply Pressure to daily life. 3. Explain Pressure in fluids. 4. Understand Buoyancy. 5. Understand and explain Archimedes Principle. 6. Applies Archimedes Principle on floating objects. 7. Understand relation between Density of a Solid and Density of a Liquid. Key Terms : 1. Thrust 2. Pressure 3. Pascal 4. Buoyancy 5. Archimedes Principle 6. Relative Density LIST OF PRACTICALS 1. To determine the density of a solid (denser than water) by using spring balance and a measuring cylinder. 2. To establish the relation between loss in weight of a solid when fully immersed in (i) tap water (ii) strongly salty water, with weight of water displaced by it taking at least two different solids. 3. To Calculate the pressure exerted by a wooden block on sand. Download full Study Note for BOARD EXAM [Given below]
Related Study material
IX Thrust and Pressure, Archimedes’ Principle, Relative Density CBSE Class 9 - Science - Chapter 10: Flotation: Notes and Quest Read MCQ: Flotation: Thrust, Pressure, Buoyancy and Density Read Thrust and Pressure, Archimedes’ Principle, Relative Density key point Read Notes : Flotation: Thrust, Pressure, Buoyancy and Density Read Physics Flotation Term-II Class IX Buoyant force Detail Study Read 9th Physics Solved Numerical Floating bodies Read |
Blog SeaRCH Link
All
Join Us For Update |
||||||||||||||||||||||||