|
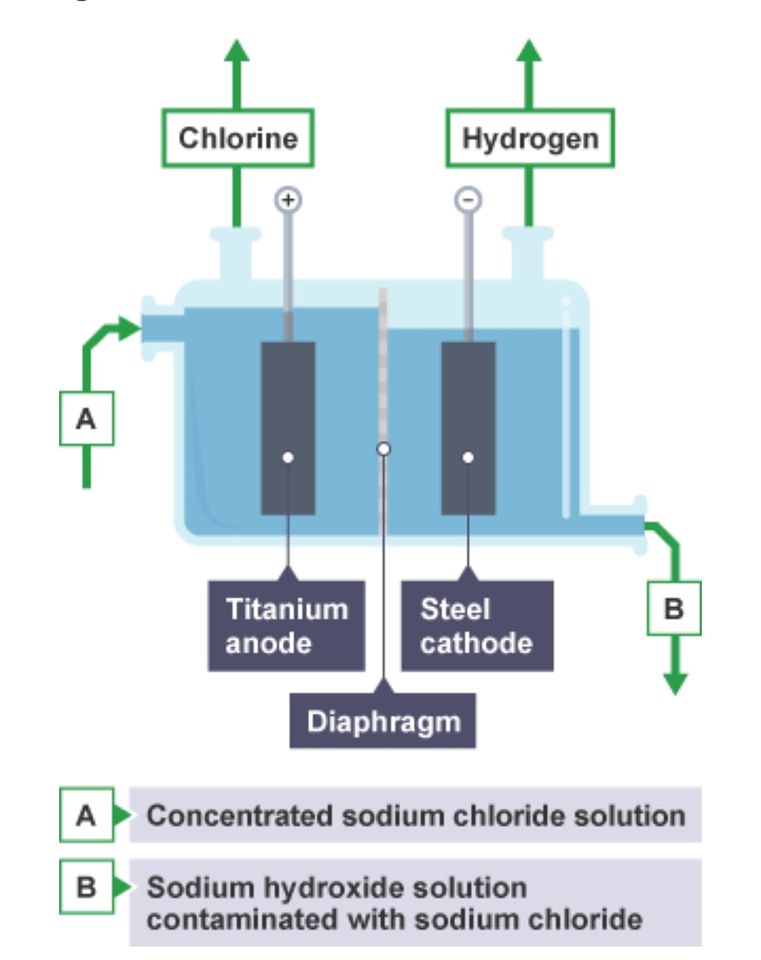
When an electric current is passed through concentrated sodium chloride solution, hydrogen gas forms at the negative electrode, chlorine gas forms at the positive electrode, and a solution of sodium hydroxide also forms.
You might have expected sodium metal to be deposited at the negative electrode. However, sodium is too reactive for this to happen so hydrogen is given off instead. During electrolysis: • hydrogen ions H+(aq) (from the water) are discharged at the negative electrode as hydrogen gas, H2(g) • chloride ions Cl–(aq) (from the dissolved sodium chloride) are discharged at the positive electrode as chlorine gas, Cl2(g) • sodium ions Na+(aq) (from the dissolved sodium chloride) and hydroxide ions OH–(aq) (from the water) stay behind - they form sodium hydroxide solution, NaOH(aq) • At the cathode (negative electrode), the H+ cations are reduced when they gain electrons: 2H+(aq) + 2e- → H2(g) • At the anode (positive electrode), the Cl- anions are oxidised when they lose electrons: 2Cl-(aq) → Cl2(g) + 2e-
0 Comments
Leave a Reply. |
Blog SeaRCH Link
All
Join Us For Update |