|
1. Gist of Lesson for Quick Revision (By JSUNIL)
2. In 1789, Lavoisier first attempted to classify the elements into two divisions namely Metals and Non-metals. 3. Mendeleev predicted the properties of those missing elements from the known properties of the other elements in the same group. On the basis of this, he named Gallium as Eka-Almunium, Germanium as Eka-Silicon and Scandium as Eka-Boron. 4. Mendeleev's periodic table helped in correcting the atomic masses of some of the elements, based on their positions in the periodic table. For example, atomic mass of beryllium was corrected from 13.5 to 9.0. Atomic masses of indium, gold and platinum were also corrected. 5. In certain pairs of elements like, Ar (40) and K (39); Co (58.9) and Ni (58.6); Te (127.6) and I (126.9) the arrangement was not justified. For example, argon was placed before potassium whereas its atomic mass is more than potassium. 6. The position of hydrogen was not correctly defined. It was placed in Group I although its properties resembled both the Group I elements (the alkali metals) and the group VII elements (the halogens). 7. Lithium, Sodium, Potassium are called Alkalies metal as they react with metal and form alkalies (Caustic solution) 8. Calcium, Strontium, Bromine are called Alkalies Earth metal as they react with metal and form salt as their oxide are alkalies in nature and exist in the Earth 9. Chlorine, Bromine, Iodine are called Halogen as their oxide are alkalies in nature and exist in the Earth 10. Alkali metal and coinage metals (Cu, Ag and Au) which differ widely in properties are placed into the same group. In 1913, an English physicist MOSELEY, as a result of his work on characteristics X-rays of elements discovered a fundamental and most accurate relation between chemical properties and atomic number of elements, known as "Modern periodic law". ACCORDING TO THE MODERN PERIODIC LAW: 1. The chemical and physical properties of elements are the periodic function of their atomic numbers". 2. The properties of elements depend upon their electronic configuration which varies with increasing atomic number in a periodic way". Period: 1. Atomic size decreases in a period. 2-Nuclear charge increases in a period. 3-Ionization potential increases in a period. 4-Electronegativity increases in a period. 5-Electropositivity and metallic character decreases in period. 6-Each period starts with Alkali metal and ends on a Noble gas. For example: 2nd period : Li .................................. Ne 3rd period : Na ..................................Ar 7-First element of each period is most reactive (electropositive), and last element is chemically inert. Group: 1-Group number of an element shows the number of electrons in the outermost shell of that element 2-Atomic size increases in group. 3-Ionization potential of elements decreases down the group. 4-Electronegativity decreases down the group. 5-Metallic character increases down the groups. On the basis of valence shell electronic configuration, periodic table is divided into four distinct blocks of elements. S-block elements All the elements in which last electron enters "ns" orbital are known as s-block elements. Elements of group I-A and II-A belong to s-block. Their valence shell electronic is ns1 to ns2. All the elements of s-block are very reactive and electropositive in nature. S-block elements are good reducing agents. They form ionic compounds. They are all metals and solid. They are low density metals. For example : Li (z=3) 1s2, 2s1 Na (z=11) 1s2, 2s2, 2p6, 3s1 Be (z=4) 1s2, 2s2 p-block elements The elements in which valence electrons enter p-orbital and this orbital is progressively filled are called p-block elements. Their valence shell electronic configuration is from ns2, np1 to ns2, np6. P-block consists of the elements of group III-A to VIII-A (zero-group). They include metals, nonmetals and metalloids. There are 30 elements in six sub-groups of p-block. d-block elements The elements in which last electron enters (n-1) d-orbital are called d-block elements or outer transition elements. Valence shell electronic configuration of these elements is ns2, (n-1) d1-10. All these elements are metals.There are four series of outer transition elements. Fourth series is incomplete. f-block elements The elements in which last electron enters (n-2) f-orbital are called f-block elements or inner transition elements. Their outer electronic configuration is ns2,(n-1)d1,(n-2) f1-14 f-block elements constitute 2 series of elements. (1) lanthanides (2) Actinides Visit fore full study: CBSE Class 10th Periodic Classification
2 Comments
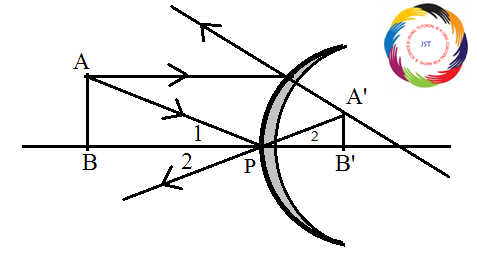
<B = <B’ and <1 = <2
D ABP ~ A’B’P [ AA - similarity] AB/A’B’ = BP/B’P ho /hi = -u/v hi /ho = -v/u Magnification = m = hi /ho = -v/u Derivation or Proof-of-Mirror formula(X) physics Class 10 CHAPTER HOW DO ORGANISMS REPRODUCE SOLVED QUESTIONS
Click here and Visit to Download File 10th CHAPTER HEREDITY AND EVOLUTION-SOLVED QUESTION SOLVED ) Click here and Visit to Download File  HOTS: (High Order Thinking Skill) Questions with Answers: Q.1. Why is variation beneficial to the species but not necessary for the individual? Ans- Variation is beneficial to the species as it enables a species for its survival. A favourable variation makes an organism to live better in a changed environment and an unfavourable variation will not. So it is not necessarily true that a variation is beneficial to the individual always. Q.2. What is the advantage of reproduction through spores in the case of Rhizopus? Ans: The spores are covered by thick walls that protect them until they come into contact with another moist surface and can begin to grow. Q.3. The simple animals such as planaria can be cut into number of pieces and each piece grows into a complex organism. What is this process known as? Ans: Regeneration Q.4) Name the unicellular organism which caused the disease known as kala-azar. Ans : Leishmania Q.5) Which process taking place in the nucleus of a cell leads to variation in the offspring during reproduction? Ans : DNA copying Q.6. What causes joining up of stock and scion in grafting technique of vegetative propagation in plants? Define the terms stock and scion. Name one positive trait each of the plant contributing scion and stock should have. Ans : The stock and scion unite due to cambial cavity. Stock is the portion on which grafting is done and it provides the roots. Scion is the portion of the plant which is grafted on the other plant and it contributes the stem. The plant contributing scion should have large sized fruits and the plant contributing stock should have deep root system. Q.7 . Which type of layering is done in Jasmine? Ans : Air Layering (Gootee) Q.8 . Where does fertilization takes place in human female? Ans : Oviduct (fallopian tube) Q.9 . Why is it said that “sexual reproduction promotes diversity of characters in the offsprings”? Ans : It is because sexual reproduction results from the fusion of two gametes coming from two different and sexually distinct individuals. This leads to variation, is necessary for evolution. Q.10. What happens if the fallopian tubes are partially blocked and the ovulated eggs are prevented from reaching the uterus? Ans : Fertilization may take place but the zygote may develop in the tube instead of uterus. Q.11 . Name the causative organism of syphilis and gonorrhoea. Ans : Treponema pallidum and Nisseria gonorrhoeae. Q.12 . Why are variation possible in progeny of sexually reproductive individuals? Ans-Variations are possible in progeny of sexually reproductive individuals because copy of DNA in newly formed cell is not identical to copy DNA of original cell. 10th NCERT Solution Intex and Exercise Questions 10th CBSE NCERT Question(1 mark) Solved 10th CBSE NCERT Question( 2 marks) Solved 10th CBSE NCERT Question( 3 marks) Solved 10th CBSE NCERT Test Paper (Solved) Click to download these files Q.What is refraction of light? What are the laws of refraction? Ans: Deviation of ray of light from its original path when it travels from one transparent homogeneous medium to another transparent homogenous medium is called the refraction of light. There are two laws of refraction :- (I ) incident ray, reflected ray and normal lie in the same plane. (ii) The ratio of sine of angle of incidence and sine of angle of refraction is constant i.e. Sin i / Sin r = n. This is also called as Snell’s law. Q. Define Refractive Index: Ans: It is the ratio of speed of light in vacuum to the speed of light in medium is called Refractive index. Q. Give the ratio of velocities of two light waves travelling is vacuum and having wave lengths 4000A0 and 8000A0. Ans: In Vacuum, light of all the wave lengths travel with the same velocity i.e. 3 X 108 m/sec. Q.. For what angle of incidence, the lateral shift produced by parallel sided glass plate is zero? Ans: For <i = 0. Q. What are the factors on which the lateral shift depends? Ans: Thickness of the refracting medium, angle of incidence and its refractive index Q. What is the value of Relative refractive index of air? Ans: One. Q. If speed of light in vacuum = 3 x 108 m/sec and Refractive index of water = 4/3, What is the speed of light in water? Ans: Refractive index of water = Speed of light in vacuum / Speed of light in water; 4/3 = (3 x 108 m/sec )/ (Speed of light in water) Speed of light in water = (3 x 108 m/sec) x ¾ = 9/4 x 108m/sec = 2.25 x 108 m/sec. Q. For the same angle of incidence in media P, Q and R, the angles of refraction are 350, 250, 150 respectively. In which medium will the velocity of light be minimum? Ans: According Snell’s law n = Sin i / Sin = C / V For given angle of incidence (i), V will be minimum, when angle of refraction <r is minimum In given data it is for medium R. Q. A coin in a glass beaker appears to rise as the beaker is slowly filled with water. Why? Ans: It happens on account of refraction of light. A ray of light starting from the coin goes from water to air and bends away from normal. Therefore, bottom of the beaker on which the coin lies appears to be raised.
|
Blog SeaRCH Link
All
Join Us For Update |
||||||