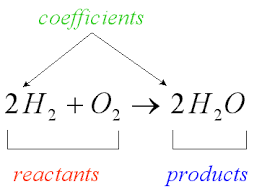
 Q. What is chemistry? Ans: Chemistry is a branch of science in which we study the composition, structure, properties and change of matter. Q. What is matter? Ans: Anything that is around us having some mass and occupies space is called matter. Q. What are the different classifications of matter? Ans: Matters are classified into element, compound and mixture. Q. What is element? Ans: Elements are the purest form of substance (matter) made up of same kinds of particles. The smallest unit of element is an atom. eg. Hydrogen, oxygen, carbon. Q. What is compound? Ans: Compound is the combination of two or more elements in ix ratio by weight. For H2O is compound in which hydrogen and oxygen are combined in 1: 8 by weight. Properties of compound are different from the constituent elements. Compound are of three type aids bases and salt Molecules are the smallest unit of compound. Q. What is mixture? Ans: A material obtained by mixing two or more element or compound in any ratio is called mixture. There are two types of mixture: a. Homogenous: A mixture having uniform composition throughout is known as Homogenous mixture. The particles of mixture cannot seen by necked eyes. eg. Milk, soda water, alloy b. Heterogenous: A mixture does not have uniform composition throughout is known as Homogenous mixture. The particles of mixture can be seen by necked eyes. eg. Air, salt in water, gun powder. Q. What is symbol? Ans: The short hand or brief representation of the name of element is called symbol. J J Berzelius given the method of writing symbols of elements using English alphabets. Q. what does a symbol represent? Ans: A symbol represent (a) Name of the element (b) one atom of elements (c) one gram atomic weight of the element Q. In what way symbol of elopement have been derived? Ans: The symbol of elopement have been derived in three ways a. Using first letter of English name for example : Hydrogen – H, Oxygen – O, Carbon – C, sulphur - S b. Using first letter along with one more letter of English name Eg. Chlorine – Cl, Helium – He, Manganese - Mn c. One or two letter of Latin name for examples: Sodium - (Natrum ) Na, Gold( Aurum) – Au, Silver (Argentinum) - Ag Q. What is Formula? Ans: The short hand representation of the name of compound is called formulae. A formula gives the idea of the name of the element and no. of element present in compounds.eg. H2, O2, Cl2, Br2, O3,P4,H2O Q. What is valancy ? Ans: The combining capacity of an element with other element to form molecules of a compound is called valency of an element. For example In H2O , The valency of Hydrogen is 1 and valancy of oxygen is two. In NH3 Valancy of nitrogen is 3 and In Al2O3 valancy of Al is 3. In CH4 valency of carbon is 4 Some element has variable valancy like Cu – 1 and 2, iron – 2 and 3, Tin – 2 and 4 , Phosphorous- 3 and 5 and sulphur- 2 ,4 and 6 Q. What are radicals? Ans: A single or groups of atoms having a positive and negative charge. eg: Na+, Cl- A radicals having only one atom is called simple radicals eg: Na+, Cl- A radicals having more than one type of atom is called simple radicals eg: NO3-1, NH4+1 The positive radicals are called basic radical and negative radicals are known as acidic radicals. Q. What is chemical reaction and equation? Ans: A chemical reaction is the process in which atoms present in substances rearrange to give new chemical combinations. The substances take part in a chemical reaction are called the reactants, and the new substances formed are called the products. Representation of chemical reaction using symbols of substances is called chemical equation. CH4 + 2 O2 → CO2 + 2 H2O 2 H2(g) + O2(g) → 2 H2O(g) 2 Al(s) + Fe2O3(s) → Al2O3(s) + 2 Fe(l) Q. What are the observations seen in chemical reaction? Ans: indications of a chemical reaction can include: Evolution of heat or light, Production of gas, Formation of precipitate or change in state and colour. Q. Why we need to balance a chemical equation? Ans: Since there is no loss or gain of atoms in a chemical reaction so, To make the equal number of atoms in reactant and product we need to balance a chemical reactions. For more study visit Class VII
14 Comments
Rangers
19/9/2015 03:32:29 pm
Worst
Reply
4/4/2023 10:35:57 am
Drfcgyhbnj xdyhbhgex ftufd
Reply
himanshee
22/9/2015 01:20:22 pm
Reply
Anasua chakraborty
22/10/2016 08:54:52 am
Good for students
Reply
Bob
21/3/2017 03:42:07 pm
My name Jeff
Reply
Chris-TG
9/6/2017 02:10:56 pm
Cmon this is not somtin i wanted plz give good stuff not q and a plz
Reply
Vartika
25/3/2018 11:03:53 pm
Quite helpful
Reply
Mihika jain
30/6/2018 03:33:35 pm
More questions like this for all chapters
Reply
24/9/2020 07:00:30 pm
some of them are so helpful, but some of them are not.OK i liked it.
Reply
Leave a Reply. |
Blog SeaRCH Link
All
Join Us For Update |


