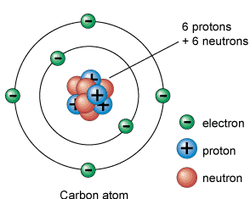
 IX Atomic Structure ( a fundamental particles) Discovery of Electron 9th Atomic Structure (Discovery of a fundamental particle protons) 9th Atoms and Molecules 9th Structure of Atoms Practice paper and sample paper based On Atomic structure Rutherford Experiment and Discovery of Nucleus Lord Rutherford (1911) and his coworkers performed an experiment. They bombarded a very thin, gold fail with Alpha particles from a radioactive source. They observed that most of the particles passed straight through the foil undeflected. But a few particles were deflected at different angles. One out of 4000 Alpha particles was deflected at an angle greater than 150. Conclusion Following conclusions were drawn from the Rutherford’s Alpha Particles scattering experiment. 1. The fact that majority of the particles went through the foil undeflected shows that most of the space occupied by an atom is empty. 2. The deflection of a few particles over a wide angle of 150 degrees shows that these particles strike with heavy body having positive charge. 3. The heavy positively charged central part of the atom is called nucleus. 4. Nearly all of the mass of atom is concentrated in the nucleus. 5. The size of the nucleus is very small as compared with the size of atom. Defects of Rutherford Model Rutherford model of an atom resembles our solar system. It has following defects: 1. According to classical electromagnetic theory, electron being charged body will emit energy continuously. Thus the orbit of the revolving electron becomes smaller and smaller until it would fall into the nucleus and atomic structure would collapse. 2. If revolving electron emits energy continuously then there should be a continuous spectrum but a line spectrum is obtained. Bohr’s Atomic Model Neil Bohr (1913) presented a model of atom which has removed the defects of Rutherford Model. This model was developed for hydrogen atom which has only proton in the nucleus and one electron is revolving around it. Postulates of Bohr’s Atomic Model The main postulates of Bohr’s Model are given below: 1. Electrons revolve around the nucleus in a fixed orbit. 2. As long as electron revolves in a fixed orbit it does not emit and absorb energy. Hence energy of electron remains constant. 3. The orbit nearest to the nucleus is the first orbit and has lowest energy. When an electron absorbs energy it jumps from lower energy orbit to higher energy orbit. Energy is emitted in the form of radiations, when an electron jumps from higher energy orbit to lower energy orbit. The unit of energy emitted in the form of radiations is called quantum. It explains the formation of atomic spectrum. Downloadable study material: Click Here Structure of atom for CBSE class 9(IX)
Solved Summative Assessment Paper Solved some important questions for coming CBSE Exam How was the neutron discovered? How was the proton discovered? How was the Electron discovered? Extra mark’s Notes, "Atomic Structure" How was the Nucleus discovered? Structure of an Atom IX Chemistry assignment-1 Structure of an Atom IX Chemistry assignment-2
0 Comments
How do you characterize sound? Intensity or Loudness
The amplitude of a wave depends on the amount of energy that it carries. A high amplitude wave carries a large amount of energy where as low amplitude wave carries a small amount of energy. The average amount of energy passing through a unit area per unit of time in a specified direction is called the intensity of the wave.When the amplitude of the sound wave increases, the intensity of the sound also increases. Sounds with higher intensities are become louder. Relative sound intensities are often given in units named decibels (dB). The Bel is the primary unit and a decibel is 1/10th of a Bel. The Bel is a ratio of the pressure of a sound to a reference pressure. The reference pressure in air is 20 microPascals (µPa). The reference pressure in water is 1 microPascal (1 µPa). Intensity: The average amount of sound power (sound energy per unit time) that transmitted through a unit area in a particular direction. The unit of intensity is watts per square meter. Full Post Read full post : 9th Sound and its Reflections IX Physics Ch. Sound Intensity and Loudness IX Physics Ch. Sound as a wave IX-VIII-Physics Refraction of Sound Explanation How does the sound produced by a vibrating object in a medium reach your ear? (Page 162 )
When an object vibrates, it vibrates the neighbouring particles of the medium. These vibrating particles then vibrate to the adjacent particles In this way, vibrations produced by an object are transferred from one particle to another till it reaches to our ear. Page 163 Explain how sound is produced by your school bell. Ans: When the bell moves backwards, it creates a region of low pressure know as rarefaction. As the bell continues to move forward and backward, it creates a series of compressions and rarefactions. This produce the sound of a bell Why sound waves are called mechanical waves? Ans: Sound waves need the material medium to vibrate and propagate. Hence, these waves are known as mechanical waves. Sound waves propagate through a medium because of the interaction of the particles present in that medium. Suppose you and your friend are on the moon. Will you be able to hear any sound produced by your friend? Sound needs a medium to propagate. Since there is no material medium on the moon due to absence of atmosphere, you cannot hear any sound on the moon. Page 166 Which wave property determines (a) loudness, (b) pitch? (a) Amplitude of sound wave determines the loudness of a sound. The amplitude of a sound directly proportional to amplitude of wave. If the amplitude of a sound is large, then the sound produced will also be loud. (b) Frequency of sound wave determines the pitch of a sound. The pitch of a sound is proportional to its frequency. High pitched sounds have high frequency Guess which sound has a higher pitch: guitar or car horn? The frequency of the vibration of a sound produced by a guitar is greater than that produced by a car horn. As we know that the pitch of a sound is directly proportional to its frequency, the guitar has a higher pitch than a car horn. Read full solution and test paper 9th_science_ncert_solution_chap.-sound Download File IX-Physics for term-II Sound CBSE Test paper Download File More use full link for physics study Physics class 9th Numerical for chapter sound CBSE TEST PAPER (Class-9)Work and Energy Numerical on work and energy for class 9 9th Question Bank chapter -Sound 9th Physics for term-II Sound NCERT Solutions Thrust and Pressure, Pressure in Fluids, Buoyancy, Archimedes’ Principle, Relative Density Key points:
1. Determine the mass of 6.022 X 10^23 number of N2 molecules.
2. Calculate the number of particles in- (i) 8 g of O2 molecules (ii) 2.5 mol of calcium atoms. 3. What is the mass of 2.5 mol of Methane? 4.Find the mass of one molecule of water. 5.Calculate the number of water molecules and number of oxygen and hydrogen atoms in a drop of water containing 0.03 mol of water. 6. Calculate the actual mass of one atom of carbon if 12 grams of Carbon contain one mole of Carbon. 7. Calculate mass of Nitrogen (N2) which contains same number of molecules as are present in 4.4 grams of Carbon-di-oxide (CO2). 8.Atomic mass of gold is 197 u. How many moles of gold are present in an ornament containing 88.65 grams of gold? 9. How many moles of SO2 have same mass as 3 moles of oxygen? 10. A glass of water contains 5 mol of water. How many molecules of water arepresent? 11. What is the mass of a formula unit of Na + C l - ? 12. How many atoms of Silver are present in a silver wire weighing 5.4 grams? 13. Calculate the ratio of molecules present in 16 g of methane and 16 g of oxygen. 14. Convert into mole. (a) 12 g of oxygen gas (b) 20 g of water (c) 22 g of carbon-dioxide. 15. Determine the number of bromide ion in 0.2 mole of Mg Br2. th Mole concept numerical problems solved-9 Download File 9th Mole concept numerical problems solved-10 Download File Class 9 Atoms and molecules solved CBSE Papers New links More Problems based on mole concept 9th Atoms and Molecules - 1. Mole Concept and Problems based on mole concept 2. Numerical Problems based on mole concept 9th Chemistry 3. Numerical based on mole concept by ChemistryAdda |
Blog SeaRCH Link
All
Join Us For Update |