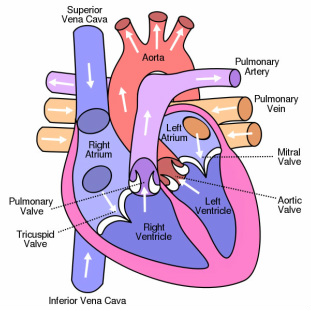
|
P. 71 Oral Questions For Formative Assessment (Living Science)
1. acetic acid and citric acid 2. Acids are corrosive in nature. Strong acids can corrode even metals like iron and aluminium. Hence, acids are not stored in metal containers. 3. litmus paper and methyl orange; acid-base indicators 4. neutralization reaction, salt 5. Organic acids are naturally occurring acids that are present in animal and plant products. They are normally weak acids. Hydrochloric acid, sulphuric acid and nitric acid are known as mineral acids. They are normally strong acids. P. 72 Oral Questions For Formative Assessment 1. Bases which are soluble in water are called alkalis. But some bases are not soluble in water, so they are not alkalis. That is why all alkalis are bases but all bases are not alkalis. 2. I will not recommend that quicklime or slaked lime be added to the soil to neutralize the acid present in it. 3. sodium hydroxide (NaOH) 4. hydrochloric acid (HCI), magnesium hydroxide is used as an an acid to neutralize the excess add in the stomach P. 74 Oral Questions For Formative Assessment 1. If we replace hydrogen of an acid with a metal, a salt is formed. 2. a salt and water 3. a. nitric acid b. carbonic acid c. hydrochloric acid d. sulphuric acid 4. a salt; sodium bicarbonate (NaHCO3) Page 74 For Formative and Summative Assessment A. 1. d 2.a 3.c 4. d 5. c 6. a 7. d 8. d 9. b B. 1. sodium chloride 2. an acid 3. red 4. false 5. nitric acid 6. lactic 7. carbon dioxide 8. neutralization 9. sulphuric add 10. alkali 11. ammonium hydroxide 12. a base 13. pale yellow 14. caustic soda 15. magnesium hydroxide 16. true 17. blue C. 1. The substance which can be used to test if a given substance is acidic or basic in nature is known as an acid-base indicatdr, for example, methyl orange. 2. lemon, antacid, common salt 3. When dilute sulphuric acid is added to zinc, hydrogen gas is produced along with zinc sulphate. 4. Dilute acids react with carbonates such as calcium carbonate (CaCO3) to form salt and carbon dioxide gas. 5. Examples of strong acids: (I) Nitric acid (ii) Sulphuric acid Examples of weak acids: (i) Lactic acid (ii) Acetic acid 6. Ant bite injects formic acid inside the skin, and thus skin irritates for some time. To get relief, a base (baking soda) is applied to neutralize the acid. The irritation ends with forming salt and water and provides relief. 7th Acids bases and Salt Living science solution Download File
7 Comments
Class 7_ Acid base and salt Solved Questions
Q. why are the sting of an ant is very painful? Answer: Ant sting contains formic acid. When an ant bites, it injects this formic acid into the skin. This causes pain. Q. Why does wasp sting should be treated with vinegar and ant sting with should be treated with baking soda? Answer: wasp stings are alkaline, and can therefore be neutralised with vinegar(acetic acid). Ant sting contains an acid , and can therefore be neutralised with base like baking soda. Q. why does an antacid tablet is taken when you suffer from acidity? Ans: An antacid tablet consists of a base like Milk of Magnesia (magnesium hydroxide). It neutralises the effect of excessive hydrochloric acid released during indigestion. Q. Why does factory waste treated before disposing into the water bodies? Ans: The wastes of many factories contain acids. If they are allowed to flow into the water bodies, the acids will kill fish and other organisms. The factory wastes are, therefore, neutralised by adding basic substances. Q. Why is calamine solution on the skin when an ant bites? Ans: Ant sting contains formic acid. When an ant bites, it injects this formic acid into the skin. This causes pain. Therefore to give relief, it can be neutralised by rubbing the ant bite with moist baking soda, which contains sodium hydrogen carbonate (NaHCO3), or with a solution of calamine, which contains zinc carbonate (ZnCO3). Q. Some acid indigestion tablet contains calcium carbonate. How do they help to make indigestion go away? why does their effect often wear off after a while? Answer: During indigestion there is a secretion of HCl in stomach. The antacid like Mg(OH)2 or NaHCO3contain base that neutralize acid and give relief. Since more acid are released in stomach and require more base so effect of indigestion tablet often wears off after a while Q. Ammonia is found in many household products, such as window cleaners. It turns red litmus blue. What is its nature? Answer: Since it turns red litmus blue, Ammonia is basic in nature. Q. Name the source from which litmus solution is obtained. What is the use of this solution? Answer: Litmus is extracted from plant lichens. It has a mauve (purple) colour when dissolved in distilled water. Q. Define acid and base? Ans: According to Arrhenius any compounds that contain hydrogen and can dissolve in water to release hydrogen ions into solution is called acid. For example, hydrochloric acid (HCl) dissolves in water as follows: HCl +H2O→ H+ (aq) + Cl-(aq) According to Arrhenius any compounds that dissolve in water to release hydroxide ions (OH-) into solution is called base. For example, sodium hydroxide (NaOH) is a base dissolves in water as follows: NaOH + H2O→ Na+(aq) + OH-(aq) According to The Brønsted-Lowry definition acid is any substance that can donate a hydrogen ion . (Under the Brønsted definition, acids are often referred to as proton donors because an H+ ion, hydrogen minus its electron, is simply a proton). According to The Brønsted-Lowry definition bases is any substance that can accept a hydrogen ion. NaOH and KOH, are bases because they can accept an H+ from an acid to form water. However, the Brønsted-Lowry definition also explains why substances that do not contain OH- can act like bases. Baking soda (NaHCO3), for example, acts like a base by accepting a hydrogen ion from an acid as illustrated below: HCl + NaHCO3 → H2CO3 + NaCl In this example, the carbonic acid formed (H2CO3) undergoes rapid decomposition to water and gaseous carbon dioxide, and so the solution bubbles as CO2 gas is released. Q. If bottle full of concentrated sulphuric acid is left open in the air by accident, the acid starts flowing out of the bottle of its own. Answer : Concentrated sulphuric acid is highly hygroscopic. It absorbs moisture from air and gets diluted. Since the volume increases, the acid starts flowing out of bottle. Q. Explain why? (a) Common salt becomes sticky during the rainy season. (b) Blue vitriol changes to white upon heating. Answer : (a) common salt contains impurity of magnesium chloride which is of deliquescent nature. When exposed to the atmosphere, it becomes moist. Therefore it becomes sticky during rainy season. (b) Blue vitriol (CuSO4.5H2O) upon heating changes to anhydrous copper sulphate (CuSO4) which is white in colour Q. Why acetic acid is called a weak acid though there are 4 'H' atoms in the molecule? Answer: Acetic acid is called a monobasic acid because only one of the four 'H' atoms of the acid is released as H+ ion in solution. Q. What happens when a base is put on turmeric paste? What will happen when acid is added to the same? Answer. When base is added to turmeric paste, then its colour changes to red. If we put an acid on the same then changes to yellow again. Q. How should tooth paste help in preventing tooth decay? Ans: When bacteria digest food in our mouths, they release weak acids, especially when we eat sugars. Our teeth have a hard covering called enamel that is weakly basic. When bacteria make our mouth acidic, the acid reacts with our enamel, removing hydroxyl ions, causing tooth decay. The active ingredient in toothpaste is a fluoride salt. When toothpaste dissolves in our mouths, negatively charged fluoride ions are released. The fluoride ions are similar to other hydroxyl ions in that they are negatively charged and can take the place of hydroxyl ions that have reacted with acid. Also, fluoride ions don't react with acid as easily as the hydroxyl ions, making enamel more resistant to decay. Q. why should acid not stored in metal container? Ans: Acids cannot be stored in metal containers as they will react with the metal, forming metal salt and liberating Hydrogen gas. Q. Which shampoo will be better for hair- One with high pH or one with low pH? Human hair has a pH balance of between 4.5 and 5.5 ie. One with low pH. This natural hair acidity prevents fungi and bacteria in the hair Q. Why should you not try to wash off turmeric stain on your cloth with soap? Ans: Turmeric is a kind of indicator which turns red when comes in contact with basic solution and as we know soap solution is basic therefore cloth get red patches. Q. How soap is prepared? Ans: soap is prepared by boiling oil or animal fat with caustic soda (Sodium hydroxide) Q. Name a base that is used to remove ink spot from cloth? Ans: NH4OH more solved Questions Acid bases and salt 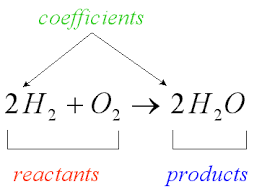 Q. What is chemistry? Ans: Chemistry is a branch of science in which we study the composition, structure, properties and change of matter. Q. What is matter? Ans: Anything that is around us having some mass and occupies space is called matter. Q. What are the different classifications of matter? Ans: Matters are classified into element, compound and mixture. Q. What is element? Ans: Elements are the purest form of substance (matter) made up of same kinds of particles. The smallest unit of element is an atom. eg. Hydrogen, oxygen, carbon. Q. What is compound? Ans: Compound is the combination of two or more elements in ix ratio by weight. For H2O is compound in which hydrogen and oxygen are combined in 1: 8 by weight. Properties of compound are different from the constituent elements. Compound are of three type aids bases and salt Molecules are the smallest unit of compound. Q. What is mixture? Ans: A material obtained by mixing two or more element or compound in any ratio is called mixture. There are two types of mixture: a. Homogenous: A mixture having uniform composition throughout is known as Homogenous mixture. The particles of mixture cannot seen by necked eyes. eg. Milk, soda water, alloy b. Heterogenous: A mixture does not have uniform composition throughout is known as Homogenous mixture. The particles of mixture can be seen by necked eyes. eg. Air, salt in water, gun powder. Q. What is symbol? Ans: The short hand or brief representation of the name of element is called symbol. J J Berzelius given the method of writing symbols of elements using English alphabets. Q. what does a symbol represent? Ans: A symbol represent (a) Name of the element (b) one atom of elements (c) one gram atomic weight of the element Q. In what way symbol of elopement have been derived? Ans: The symbol of elopement have been derived in three ways a. Using first letter of English name for example : Hydrogen – H, Oxygen – O, Carbon – C, sulphur - S b. Using first letter along with one more letter of English name Eg. Chlorine – Cl, Helium – He, Manganese - Mn c. One or two letter of Latin name for examples: Sodium - (Natrum ) Na, Gold( Aurum) – Au, Silver (Argentinum) - Ag Q. What is Formula? Ans: The short hand representation of the name of compound is called formulae. A formula gives the idea of the name of the element and no. of element present in compounds.eg. H2, O2, Cl2, Br2, O3,P4,H2O Q. What is valancy ? Ans: The combining capacity of an element with other element to form molecules of a compound is called valency of an element. For example In H2O , The valency of Hydrogen is 1 and valancy of oxygen is two. In NH3 Valancy of nitrogen is 3 and In Al2O3 valancy of Al is 3. In CH4 valency of carbon is 4 Some element has variable valancy like Cu – 1 and 2, iron – 2 and 3, Tin – 2 and 4 , Phosphorous- 3 and 5 and sulphur- 2 ,4 and 6 Q. What are radicals? Ans: A single or groups of atoms having a positive and negative charge. eg: Na+, Cl- A radicals having only one atom is called simple radicals eg: Na+, Cl- A radicals having more than one type of atom is called simple radicals eg: NO3-1, NH4+1 The positive radicals are called basic radical and negative radicals are known as acidic radicals. Q. What is chemical reaction and equation? Ans: A chemical reaction is the process in which atoms present in substances rearrange to give new chemical combinations. The substances take part in a chemical reaction are called the reactants, and the new substances formed are called the products. Representation of chemical reaction using symbols of substances is called chemical equation. CH4 + 2 O2 → CO2 + 2 H2O 2 H2(g) + O2(g) → 2 H2O(g) 2 Al(s) + Fe2O3(s) → Al2O3(s) + 2 Fe(l) Q. What are the observations seen in chemical reaction? Ans: indications of a chemical reaction can include: Evolution of heat or light, Production of gas, Formation of precipitate or change in state and colour. Q. Why we need to balance a chemical equation? Ans: Since there is no loss or gain of atoms in a chemical reaction so, To make the equal number of atoms in reactant and product we need to balance a chemical reactions. For more study visit Class VII
|
Blog SeaRCH Link
All
Join Us For Update |