|
POINTS TO REMEMBER:
1) ECOLOGY – The study of the interaction of living organisms with each other and their surroundings is called ecology. 2) ENVIRONMENT – Everything that surrounds organisms and influences its life. a) Biotic components of environment --- The living organisms .e.g. Plants and animals. b) Abiotic components of environment --- The nonliving components like water, air, light, etc. It is the sum total of all external conditions and influences that affect the life and the development of organisms i.e.,it includes all the biotic and abiotic factors. 3) ECOSYSTEM – All interacting organisms in an area together with the nonliving constituents of environment. (Functional unit of an environment) Functions of ecosystem:- a) Flow of energy b) Cycling of nutrients(bio-geo chemical cycles). 4) PRODUCERS --- They make the energy from sunlight available to the rest of the ecosystem. 5) CONSUMERS --- Animals can not manufacture their own food. They are called consumers. 6) BIODEGRABLE ---- Substances that are broken down by the action of bacteria or other saprophytes. e. g. – Paper. 7) NONBIODEGRABLE --- Substances that are not broken down by the action of bacteria or saprophytes. e. g. Plastic. 8) FOOD CHAIN ---- The process of one organism eating the other. GRASS -> GRASSHOPPER -> FROG -> SNAKE Importance of food chain 1.It helps in transfer of food energy from one organism to another. 2.It is a pathway for the flow of energy. 3. It helps in understanding the interdependence amongst different organisms. Trophic levels are the various steps in the food chain . Producers --- first trophic level Herbivores --- second trophic levels Carnivores --- third trophic level Top carnivores --- fourth trophic level 9) FOODWEB ----- It is a network of food links between populations in a community. 10) FLOW OF ENERGY -Ten percent law Energy available at each successive trophic level is 10 % of the previous level. (Lindeman 1942) The law states that, “ only a 10 % amount of the total available energy is transferred from one trophic level to the next. The rest 90% of energy is used up or lost to the surrounding.” 11) BIOLOGICAL MAGNIFICATION --- Progressive accumulation of nonbiodegradable waste at various trophic levels of food chain. 12. Differences between food chain & food web Food chain 1. The process of eating & being eaten to transfer food energy. 2. It forms a part of food web. 3. It has limited populations. Food web 1. It is a system of interconnected food chains. 2. It contains many food chains. 3. It has several populations of different species. 13. HOW DO OUR ACTIVITIES AFFECT THE ENVIRONMENT? Two major problems:- a) Depletion of ozone layer b) Disposal of wasteGood ozone and bad ozone The thick blanket of ozone layer in the atmosphere which forms a protective cover & prevents UV radiation from reaching the earth‘s surface. This is called good ozone and is found in the stratosphere. Ozone when present in the troposphere act as a pollutant . This harmful ozone is called bad ozone. 14. Depletion of ozone layer : The ozone layer protects all the organisms from the harmful ultra violet rays. Ozone depleting substances: CFCs, N2O, CH4 , CCl4 These chlorine containing compounds are used in: Aerosols, solvents, refrigerants and fire extinguishers. Ozone hole developed over Antartica. 15. Effects of ozone depletion on human health- Due to depletion of ozone layer UV radiations reaches the earth and cause: i) skin cancer ii) increased chances of cataract iii) suppression of immune system . Managing the garbage we produce 16. Reasons for large production of garbage i) Improvement in life style ii) Changes in attitude (more things become disposable) iii) Population explosion iv) Changes in packaging(non -biodegradable) More topics to read FORMATIVE ASSIGNMENT - III 10th Our Environment 10th Our Environment HIGHER ORDER THINKIN SKILLS (HOTS) QUESTIONS 10th physics Chapter - 15 Our Environment POINTS TO REMEMBER
0 Comments
Human eye can be treated as an optical instrument. Light rays coming from the object to be seen enter the eye through Cornea and fall on the eye lens through the pupil of the eye.
Eye lens being double convex lens forms a real inverted and smaller image of the object on the retina. The retina contains numerous light sensitive cells which are activated by the light falling on the retina and generate electrical signals which are sent to brain via optic nerve and the brain processes this information and we perceive objects as they are. The human eye has the following parts:- (a) Cornea:-The transparent spherical membrane covering the front of the eye. (b) Iris:-The coloured diaphragm between the cornea and lens. (c) Pupil:-The small hole in the iris. (d) Eye lens:-Its is a transparent lens made of jelly like material. (e) Ciliary muscles:-These muscles hold the lens in position. (f) Retina:-The back surface of the eye. (g) Blind spot:-The point at which the optic nerve leaves the eye. An image formed at this point is not sent to the brain. (h) Aqueous humour:-A clear liquid region between the cornea and the lens. (i) Vitreous humour:-The space between eye lens and retina is is filled with another liquid called Vitreous humour. Persistence of vision: The image of an object seen persists on the retina for 1/16 second even after the removal of the object. This continuance of sensation of eye for some timed is called persistence of vision. Colour blindness: It is said to occur when a person cannot distinguish between colours though his vision may otherwise be normal. Read more » The electronic configuration of carbon is K=2, L=4. It has four electrons in the valence shell and belongs to group IV A (group 14) of the periodic table.
Carbon is a non metal. In nature, it occurs in its pure form as diamond and graphite. When fuels burn, the carbon in them reacts with oxygen to form carbon dioxide. Carbon circulates through air, plants, animals and soil by means of complex reactions. This is called carbon cycle.In the beginning of 19th century scientists classified the compounds of carbon into twotypes, based on their source of occurrence: i) Inorganic compounds (obtained from non living matter)ii) Organic compounds (obtained from living matter, such as plant and animal sources) however the basis of classification was subjected to alteration after wohler synthesis FRIEDRICH WOHLER : A creator of revolution in ORGANIC CHEMISTRY The term organic chemistry was used by the Swedish chemist Berzelius. This refers to the chemistry of living things. However, the German chemist Wohler succeeded in creating an organic compound (urea) from an inorganic compound (ammonium cyanate) in his laboratory. This has dealt a severe blow to the vital force theory (a theory of life process). MODERN DEFINITION OF ORGANIC CHEMISTRY DOWNLOAD FULL NOTES AND MORE
Imortant term & condition:
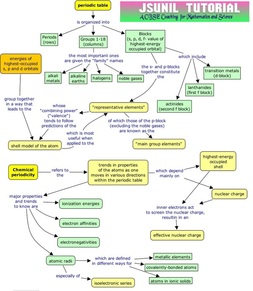
Classification of elements: the arrangement of elements in such a manner that elements with similar properties are grouped together while elements with dissimilar properties are separated. EARLY ATTEMPTS OF CLASSIFICATION OF ELEMENTS 1. Lavoisier’s classification of elements: In 1789, Lavoisier first attempted to classify the elements into two divisions namely Metals and Non-metals. However this classification was not satisfactory as there were many exceptions in each category. 2. Dobereiner’s classification of elements: In 1817, Johann Wolfgang Dobereiner grouped three elements into what he termed triads.In each case, the middle element has an atomic mass almost equal to the average atomic masses of the other two elements in the triad.For example, elements like lithium, sodium and potassium have atomic masses 7, 23 and 39 respectively. They are grouped together into a triad as,Li (7) Na (23) K (39)Here the atomic mass of sodium is the average of atomic masses of lithium and potassium. Limitation of Dobereiner’s law Only three triads were identified from the element known at that time. Hence, this classification was not useful. Newland’s law of octaves: Read more » Q. 1. How much percentage of earth’s crust constitutes carbon element ?
Ans. The earth’s crust has only 0·02% of carbon in the form of minerals. Q. 2. What do you mean by covalency ? Ans. Covalency is defined as the number of electrons shared by an atom. Q. 3. What is covalent bond ? Ans. A bond formed by the sharing of electrons is called covalent bond. Q. 4. What is functional group ? Ans. A functional group is an atom or a group of atoms that characterizes the chemical properties of an organic compound. Q. 5. What is organic chemistry ? Ans. The branch of chemistry that deals with study of compounds of carbon except for oxides of carbon, carbonate and hydrogen carbonate salts is called organic chemistry. Q. 6. What are Hydrocarbons ? Ans. These are the compunds of carbon and hydrogen are called hydrocarbon. Q. 7. What are saturated carbon compounds ? Ans. The compunds of carbon in which carbon-carbon atoms are linked by single covalent bond are called saturated carbon atoms; Alkanes. Q. 8. What are unsaturated carbon compunds ? Ans. The compounds of carbon in which carbon-carbon atoms are linked by multiple bonds i.e. double or triple bonds are called unsaturatedcarbon compounds, e.g : Alkenes or Alkynes. Q. 9. Define Allotropy. Ans. The phenomena by virtue of which an element can exist in two or more than two forms having similar chemical properties but different physical properties is called allotropy. Q. 10. What is vinegar ? Ans. Vivegar is 5–8% solution of acetic acid in water. SHORT ANSWERS TYPE QUESTIONS 2 Marks each 1.What is combustion ? Ans. It is the process of burning of a substance in the presence of oxygen to produce carbon dioxide along with heat and light.Combustion reactions are also called oxidation reactions. e.g. : CH4 + O2 ——> CO2 + H2O + Heat and light 2. Write two differences between oils and fats. Ans. Oils : 1. Oils are liquid at room temperature 2. Oils contaning unsaturated fatty acids. Fats : 1. Fats are solid at room temperature. 2. Fats containing saturated fatty acids. 3. Define Esterification ? Ans. It is the process of formation of ester by the reaction between an alcohol and an acid is called esterification. e.g. : Reaction between ethanol and ethanoic acid in presence of conc. H2SO4. CH3COOH + C2H5OH ——> CH3COOC2H5 + H2O Ethanoic acid Ethanol Ethylethanoate Water 4. What is saponification ? Ans. It is the alkaline hydrolysis of esters to give back the carboxylic acid and alcohol. As it is used in the prepation of soap, it is called saponification. CH3COOH + NaOH ——> CH3COONa + C2H5OH Ethanoic acid Sodiumhydroxide Sodiumethanoate Ethanol 5. Which of the following hydrocarbons undergo addition reactions : C2H6, C3H8, C3H6, C2H2and CH4. Ans. Unsaturated hydrocarbons, i.e., Alkenes and alkynes undergo addition reactions. Alkenes and alkynes have general formula CnH2n and CnH2n -2. Thus compounds — C3H6 (propene) and, C2H2 (ethyne) ; undergo addition rections. 6. How can you differentiate saturated and unsaturated Hydrocarbon on the basis of burning behaviour ? Ans. On burning, saturated hydrocarbons give a clean flame whereas unsaturated Hydrocarbons on heating give a yellow flame with lots of black smoke. 7. Write two ill-effects of drinking excess of alcohol ? Ans. (1) Alcohol causes addiction and makes person dependent on it. (2) If consumed in large quantities, it may cause death by damaging liver. 8. What is denatured alcohol ? Ans. Alcohol which is made unfit for the drinking purposes is called denatured alcohol. It is ethyl alcohol (C2H5OH) mixed with poisonous substances like methanol, copper sulphate, etc. 9. Give two advantages of synthetic detergents over soaps ? Ans. (1) Synthetic detergents can be used with hard water also whereas soaps cannot with hard water. (2) Synthetic detergents are more soluble and have better cleansing action than soaps. 10. What are substitution reactions ? Ans. It is a type of reaction in which a chemical reagent (substituent) displaces the another atom or group (leaving group) in a compound, e.g., Substitution of Hydrogen of Methane by chlorine CH4 + Cl2 --Sunlight ----------> CH3Cl + HCl Methane Chlorine Methyl Chloride Hydrogen Chloride or Chloramethane SHORT ANSWERS TYPE QUESTIONS 3 Marks each 1. Differentiate between diamond and graphite. Ans. Diamond : 1. Diamond has 3-dimensional network structure. 2. Each carbon in diamond is bonded to four other carbon atoms. 3. No free electron is left. 4. It is a bad conductor of electricity. 5. It is hard. Graphite : 1. Graphite has hexagonal sheet layer structure. 2. Each carbon atom in graphite is bonded to three other carbon carbon atoms. 3. Each carbon has one free electron with it. 4. It is a good conductor of electricity. 5. It is soft and slippery. 2. How is combustion different from pyrolysis ? Ans. Combustion means heating a compound in the presence of excess or limited supply of air, e.g., combustion of methane gives carbon dioxide and water. CH4 + O2 ——> CO2 + H2O + Heat and light. Pyrolysis means the decomposition of a hydrocarbon into its element on strong heating in the absence of air, e.g. : Pyrolysis of methane gives carbon and hydrogen. CH4 ------100°C-------------> C + 2H2 In abesebce of air 3.Discuss the method of preparation of soap in the laboratory. Ans. In laboratory, soap is prepared by heating of equal volumes of castor oil and concentrated sodium hydroxide solution taken in a beaker. A paste of soap is formed by constant stirring during heating. Most of the soap separates out but some of it remains dissolved in solution. NaCl is then added to precipated out all the soap from equeoussolution. On cooling the solution, solid soap separates out. Glycerl Oleate + Sodium ---Heat -> Sodium Oleate + Glycerol Glycerol is obtained as a byproduct in this process. 4. Write five ill effects of alcohol drinking. Ans. (1) Alcohol is an intoxicant and its drinking result lose in the sense of discrimination. (2) It ruins the health of the person by damaging liver and making his brain dull. (3) Alcohol drinking by the head of a family worsens the economic condition of the family. (4) The alcohol drinking by the head of a family has very bad effect on the psychological development of the children. (5) Drinking adulterated alcohol causes blindness or even death. 5. Differentiate between soaps and synthetic Detergents. Ans. Differences between soaps and synthetic detergents : Soaps : 1. They are the sodium salts of the long chain carboxylic acids. The ionic group in soaps is – COO Na 2. Soaps have relatively weak cleansing action. 3. Soaps are prepared from animal fats or vegtable oils. 4. They are not suitable for washing clothes in Hard water. 5. Soaps are biodegradable. Synthetic Detergents : 1. Synthetic detergents are the sodium salts of long chain benzene sulphone acids or long chair alkyl hydrogen sulphate. The ionic group in a synthetic detergent is — SO3Na or —SO4—Na+. 2. Synthetic detergents have a strong cleasing action. 3. Synthetic detergents are prepared from the hydrocarbons. 4. Synthetic detergents can be used for washing clothes even in Hard Water. 5. Synthetic detergents are non-biodegradable. 6. Differentiate between ionic compounds and covalent compounds. Ans. Difference between ionic compounds and covalent compounds : Ionic compounds : 1. Ionic compounds have constituents particles called ions. 2. In these compounds the ions of opposite charge are held together by strong electrostatic forces of attraction. 3. They have high melting and boiling points. 4. These conduct electricity in solution or in molten state. Covalent compounds : 1. Covalent compounds have constituent particles called molecules. 2. In these compounds the molecules are held together by weak attractive forces. 3. They have low melting and boiling points. 4. These are largely non-conductor. 7. Give some important properties of ethanol (ethyl alcohol). Ans. (1) Ethanol (C2H5OH) is highly soluble in water. (2) It is a colourless liquid at room temperature. (3) It has very low melting point (156 K). (4) It has low boiling point (351 K or 78ºC). (5) It is one of the main components of alcoholic drinks. (6) It is a good organic solvent. (7) Ethanol alcohol reacts with Na metal vigorously to evolve H2 gas. 2C2H5OH + 2Na ——> 2C2H5ONa + H2 (g) Sodium ethoxide (8) It burns with blue flame in the presence of O2 of air. This combustion is an oxidation process. C2H5OH + 3O2 -Combustion ----->2CO2 + 3H2O + Heat and light 8. Give five main advantages of synthetic detergents over soaps. Ans. Following are the advantages of synthetic detergents : a. Synthetic detergents can be used even with hard water but soaps cannot be used with hard water. b. Synthetic detergents have a stronger cleansing action than soaps. c. Synthetic detegents are more soluble in water than soaps. d. Synthetic detergents are prepeard from the hydrocarbons white soaps are prepared from from animal fat or vegetable oil which leads to their deficiency for human consumption. e. Synthetic detergents can be used even in acidic solution but not the soaps. 9. Write important uses of (a) ethanol and (b) ethanoic acid. Ans. (a) Uses of ethanol : (1) Ethanol (ethyl alcohol) is used for making alcoholic drinks like wines, whiskies, beer, etc. (2) It is used for making wood polish. (3) It is used for making some medicines like cough syrups, tincture iodine and some tonics. (4) It is used for making antifreeze material for cooling engines of vehicles. (b) Uses of ethanoic acid : (1) Dil. solution of ethanoic acid (acidic acid) in water is called vinegar, which is used as a preservative of pickles. (2) It is used as a useful reagent in the chemical laboratories. (3) It is used for making many C– compounds. (4) It is used for making esters which are used as flavouring agents and perfumes. 10. What happens when ethanol reacts with (i) sodium (ii) potassium permanganate solution. Ans-(i) with sodium metal ,sodium ethoxide formed. 2CH3CH2OH+2Na ------------> 2CH3CH2ONa + H2 (ii) With alkaline potassium permanganate, ethanol is oxidised to ethanoic acid. CH3CH2OH+2[O] --------> KMnO4 ---Dil.H2SO4 -----> CH3COOH + H2O Posts Related to Chapter 10th Carbon and its Compounds View/Study Carbon compounds - Points to remember and Important questions paper Naming of Hydrocarbons -No.1 Site For Learning Chemistry CBSE ADDA: Short types Questions class 10 Carbon and its Compounds 10th Chemistry Carbon and its Compounds Notes-1 Chemistry X | Carbon and its Compounds | Short type important questions Carbon and its compounds CBSE Solved Test Paper Carbon and its compounds CBSE Test Paper CARBON AND ITS COMPOUNDS QUICK PREPARATION Download Link |
Blog SeaRCH Link
All
Join Us For Update |