0 Comments
Carbon and its compounds F.A-III QUIZ and True or False Questions Paper for class 10 Chemistry2/10/2012 Carbon and its compounds QUIZ
1. Name the product of Dehydration of ethanol with conc. H2SO4 at 443 K ? 2. What is the common name of ethanoic acid? 3. Name the allotropic form of carbon with the structure of football? 4. Name the element which shows the property of catenation to the maximum extent? 5. Name the allotropic form of carbon which contain carbon- carbon double bond? 6. Name the allotropic form of carbon which is good conductor of electricity? 7. What is the general formula of alkane? 8. Out of ethanol and methanol, which is poisonous in nature? 9. Name the class of organic compound with the fruity odour? 10. What is the molecular formula of Benzene? ANS: 1. Ehene 2.Acetic Acid 3. Flullerene 4. Carbon 5. Graphite 6.Graphite 7. CnH2n+2 8. Methanol 9. Ester 10. C6H6 Q. State whether the following statement are True or False 1. Any number of atoms can be linked by Covalent bond _____ 2. The central atom in a covalent molecule has always 8 electrons after sharing ______ 3. Covalent compound are usually water soluble ______ 4. Covalent compound has generally low melting and boiling points ______ 5. Diamond is good conductor of electricity ______ 6. Each family is characterized by functional group which is an atom or group of atoms 7. Ethanoic acid and ethanol can be distinguished by litmus test as well as by sodium bicarbonate test _ 8. Soaps are water soluble while detergents are water in- soluble ______ 9. Mineral acids are stronger acids than carboxylic acids. ______ 10. Dilute alkaline KMnO4 is an oxidising agent ______ Ans: 1. False 2.True 3.False 4. True 5. False 6. True 7. True 8.False 9.True 10.True For more Paper visit Carbon and its compound Downloads 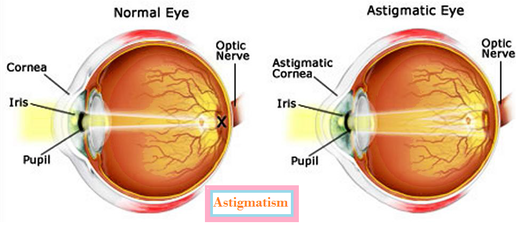 Q. The near point of a hypermetropic eye is 1 m. What is the power of the lens required to correct this defect? Assume that the near point of the normal eye is 25 cm. Answer: In the problem it is given that the near point of the normal eye is 25 cm, hence u = - 25 cm. The near point of a hypermetropic eye is 1 m . It is the nearest point up to which the hyper me tropic eyes can see object. So to correct hypermetropic eyes of this defect, person has to use Spectacles with convex lens of suitable focal length. This lens form virtual image of nearby object at the near point of hypermetropic eye. so image distance is 1m=100cm v= near point of hypermetropic eye = -1m = -100 cm u = near point of normal eye = -25cm. 1/f = 1/v -1/u = -1/100 + 1/25 = (-1 + 4)/100 =3/100 f = 100/3 cm = 1/3 m Power = 1/f = 1/(1/3) =3 dioptre Q. The far point of a myopic person is 80 cm in front of the eye. What is the nature and power of the lens required to correct the problem? Answer: This defect is corrected by concave lens. For myopic personthe object distance is ¥ since the far point this eyes is 80 cm. So to correct myopic eyes of this defect, person has to use Spectacles with concave lens of suitable focal length. This lens form virtual image of distant object at the far point of myopic eye. So image distance is 80 cm so applying lens formula 1/v - 1/u = 1/f v = - 80 cm u = infinity=0 -1/80 + 1/o = 1/f 1/f = -1/80 f = -80 cm or -0.8 m power is = -1/0.8 = -10/8 = -1.25 D So it would be a concave lens as it has a negative power Why is the colour of the clear Sky Blue? The blue light has lest velocity. Thus, when sunlight passes through the atmosphere, the fine particles in air scatter the blue colour (shorter wavelengths) more strongly. Hence the colour of the clear Sky appears blue. If there is no atmosphere, there would not have been any scattering. Then, the sky would have looked dark. Explain reason for the reddish appearance of the Sun at the sunrise or sunset. At the sunrise or the sunset, Light from the Sun has to cover larger distance in the earth’s atmosphere before reaching our eyes. Since red colour light has longer wave length deviate lesser then other colours light and reach to our eyes. What is Persistence of vision? Ans: The impression of image is remains on retina for about 1/16th of second. This is used during cinematography. The sequence of still picture taken by movie camera is projected on the screen at he rate of 24 or more images per second so that images merge in one another on screen . What is astigmatism? In astigmatism a person can see only in one plane either horizontal or vertical. When light rays enter the eye they do not focus correctly on the retina, resulting in a blurred image. Astigmatism may also be caused by an irregularly shaped lens, which is located behind the cornea. 10th chapter wise solution PERIODIC PROPERTIES
in a period the number of valence electrons and the nuclear charge increases from left to right. It increases the force of attraction between them. In a group the number of filled shells increases and valence electrons are present in higher shells. This decreases the force of attraction between them and the nucleus of the atom. These changes affect various properties of elements and they show gradual variation in a group and in a period and they repeat themselves after a certain interval of atomic number. Such properties are called periodic properties. Atomic radius What happens to atomic radii in a group and period and why? Atomic radius is the distance between the centre of atom and the outermost shell. In a period, atomic radius generally decreases from left to right. 2nd Period Li Be B C N O F 155 112 98 91 92 73 72 3rd Period Na Mg Al Si P S Cl 190 160 143 132 128 127 99 In a period there is a gradual increase in the nuclear charge. Since valence electrons are added in the same shell, they are more and more strongly attracted towards nucleus. This gradually decreases atomic radii. Atomic radii increase in a group from top to bottom. Element Atomic radius Element Atomic radius Li 155 F 72 Na 190 Cl 99 K 235 Br 114 Rb 248 I 133 As we go down a group the number of shells increases and valence electrons are present in higher shell and the distance of valence electrons from nucleus increases. Also, the number of filled shells between valence electrons and nucleus increases Both the factors decrease the force of attraction between nucleus and valence electron. Therefore, atomic size increases on moving down a group. Ionization energy Negatively charged electrons in an atom are attracted by the positively charged nucleus. For removing an electron this attractive force must be overcome by spending some energy. The minimum amount of energy required to remove an electron from a gaseous atom in its ground state to form a gaseous ion is called ionization energy. It is measured in unit of kJ mol-1. It is a measure of the force of attraction between the nucleus and the outermost electron. Stronger the force of attraction, greater is the value of ionization energy. It corresponds to the following process: If only one electron is removed, the ionization energy is known as the first ionization energy. If second electron is removed the ionization energy is called the second ionization energy. Ionization energy decreases in a group from top to bottom This is because the force of attraction between valence electrons and nucleus decreases in a group from top to bottom. The ionization energy increases in a period from left to right. This is because the force of attraction between valence electron and nucleus increases in a period from left to right. Electron affinity: The tendency of element to gain an additional electron. This ability is measured by electron affinity. It is the energy change when an electron is accepted by an atom in the gaseous state. Electron affinity is assigned a positive value when energy is released during the process. Greater the value of electron affinity, more energy is released during the process and greater is the tendency of the atom to gain electron. In a group, the electron affinity decreases on moving from top to bottom as less and less amount of energy is released. In a period, the electron affinity increases from left to right, as more and more amount of energy is released Electro negativity Electro negativity is relative tendency of a bonded atom to attract the bond-electrons towards it self. It just compares the tendency of various elements to attract the bond-electrons towards themselves. Electro negativity decreases in a group from top to bottom. Electro negativity increases in a period from left to right. Electro negativity is related to ionization energy. Electrons with low ionization energies have low electronegativity because their nuclei do not exert a strong attractive force on electrons. Elements with high ionization energies have high electronegativity due to the strong pull exerted on electrons by the nucleus. Oxides and its nature: Metals react with oxygen to form oxides by loss of electrons. These oxides on dissolution in water form bases. Reactivity of elements: Down the group reactivity of metals increases as the tendency to lose electrons increases due to increased atomic size. Reactivity of non- metals decreases down the group because of the increased atomic size and the tendency to gain electrons decreases. On moving across the period, the reactivity first increases due to the decrease in the metallic character and increase in non metallic character. Metallic and Non-metallic Properties The metallic character of an element is expressed in terms of its electron releasing tendency while non-metallic character in term of electron accepting tendency. In group metallic character of the element increases down the group due to increasing atomic size or because outermost electrons are farther away from the nucleus .So they can be easily lost. In periods metallic character of the element decreases along a period due to decrease in atomic size along a period or outermost electrons are closer to nucleus. So they can not be easily lost. Periodic Properties of the Elements –X Chemistry by JSUNIL Periodic Properties Check your understanding How to identify the group and period of element ?
Electronic configuration is the arrangement of electrons in an atom in different shells and sub-shells. The modern periodic table has elements arranged according to their electronic configurations.From left to right i.e. in a period,the arrangment is such that each subsequent electron added is to the same shell. Electronic Configuration is the Period e.g Configuration of Li atomic number. 3, is 1s2 ,2s1 Configuration of Be atomic no. 4, is 1s2 ,2s2 Configuration of B atomic no. 5, is 1s2 ,2s2,2p1 Configuration of C atomic no. 6, is 1s2 ,2s2 ,2p2 and so on till Neon,the last element. This addition of each subsequent electron results in a completely new configuration and as such the chemical properties of the subsequent element change gradually..At the end of each row, a drastic shift occurs in chemical properties. Electronic Configuration is the Group From top to down in a column,i.e.in a group, each subsequent electron is added to the new shell,but in same sub-shell. e.g. In the 1st group, Configuration of H atomic number.1,is 1s1 Configuration of Li atomic no. 3, is 1s2 ,2s1 Configuration of Na atomic no. 11, is 1s2 ,s2,2p6 ,3s1 Configuration of K atomic no. 19, is 1s2 ,2s2 ,2p6,3s2 ,3p6,4s1 and so on till Neon,the last element. The number of valence shell electrons in an atom would determine the way it will interact with other atoms, and therefore determine its chemical properties. Further study visit10th Periodic Classification of Elements (10 pages) |
Blog SeaRCH Link
All
Join Us For Update |


