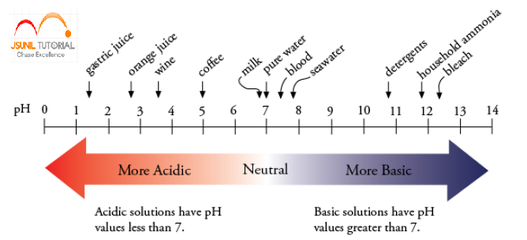
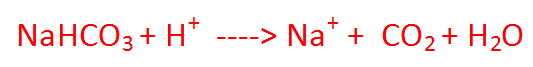 Question: The pH of hydrochloric acid solution is 3. Does it mean that it has only hydronium ions? If not, how are OH- ions generated? Answer: It is found that for any aqueous solution (neutral, acidic or basic), the product of H+ ion concentration and OH- ion concentration is always constant and equal to 10-14 at 25°C.This is called ionic product of water(Kw) Kw= [H+] [OH - ] = 10-14 Here The pH of hydrochloric acid solution is 3. Let OH- ion concentration = p [H+] [OH - ] = 10-14 1.0 × 10-3 x p = 10-14 p = [10-14]¸[ 1.0 × 10-3] =1.0 × 10-11 Thus for a solution if H+ concentration is known, OH- ion concentration is fixed (because their product is always constant).Hence we express the acidic, basic or neutral character of any solution by expressing its H+ ion concentration in terms of pH where p stands for potenz ie power.  Question: What is aqua-regia ? Answer: A mixture of concentrated nitric and hydrochloric acids. It is a highly corrosive liquid able to attack gold and other resistant substances. Question: Plaster of Paris should be stored in a moisture-proof container. Explain why? Answer: Plaster of paris should be stored in moisture proof container because Plaster of paris is powdered form of gypsum with less water of crystallization. But when we keep in open it will react with water to for hard solid mass Gypsum. CaSO4. 1/2H2O + 3/ H2O ------- > CaSO4. 2H2O Question: Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. Amount and concentration taken for both the acids are same. In which test tube will the fizzing occur more vigorously and why? Answer:-In test tube A fizzing occur more vigorously. Because HCl is stronger acid than acetic acid. Hence, HCl liberates hydrogen gas more vigorously, which causes fizzing more vigorously. What will happen if heating is not controlled while preparing Plaster of Paris from gypsum?19/6/2014  Baking soda is a single compound which has sodium hydrogen carbonate which is alkaline (basic) in nature . Baking powder is a mixture of sodium hydrogencarbonate and a mild edible(eatable) acid such as tartaric acid. When baking powder mixes with water, then the sodium hydrogencarbonate reacts with tartaric acid to evolve carbon dioxide gas which gets trapped in the wet dough and bubbles out slowly making the cake to rise and hence 'soft and spongy'.The equation which takes place can be shown as: |