|
Carbon cannot reduce the oxides of sodium, magnesium, calcium, aluminium, etc., to the respective metals. This is because these metals have more affinity for oxygen than carbon. These metals are obtained by electrolytic reduction. For example, sodium, magnesium and calcium are obtained by the electrolysis of their molten chlorides. The metals are deposited at the cathode (the negatively charged electrode), whereas, chlorine is liberated at the anode (the positively charged electrode). The reactions are : At cathode (Na⁺) + (e⁻) → Na ; At anode (2Cl⁻) → Cl2 + (2e⁻) Similarly, aluminium is obtained by the electrolytic reduction of aluminium oxide. Electrolytic reduction of copper involves the extraction of copper metal from copper ions using electricity. Here's a detailed explanation of the process:
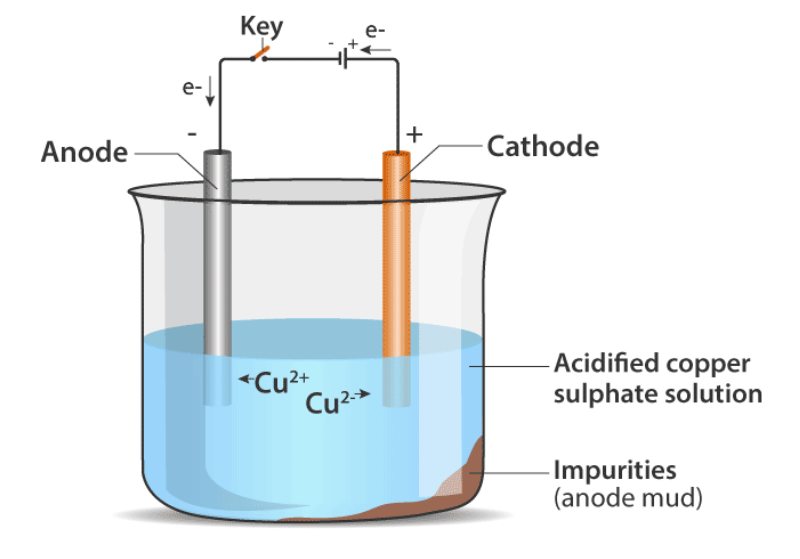
Electrolytic purification of metal is a process used to remove impurities from a metal by using electricity. Let's explore this process with an example of the electrolytic purification of copper. Here's how the electrolytic purification of copper works:
Electrolytic refining and electrolytic reduction are two distinct electrochemical processes, each serving a specific purpose in the extraction and purification of metals. Let's delve into the detailed differences between the two: Electrolytic Refining:
Here are some questions related to electrolytic refining and electrolytic reduction suitable for CBSE Class 10: Electrolytic Refining Questions:
Answers:
Case Study Questions: Here are case study questions based on the topics of electrolytic refining and electrolytic reduction, including two questions worth 1 mark each and two questions worth 2 marks each: Case Study: Electrolytic Processes in Metal Production Background: A metallurgical plant is using electrolytic processes for the production of high-purity metals. Two specific processes are electrolytic refining and electrolytic reduction. Question 1 (1 mark):
Here are assertion and reason-based questions related to the topics of electrolytic refining and electrolytic reduction: Assertion 1: Electrolytic refining is a process used to obtain high-purity metals from their ores. Reason 1: In electrolytic refining, the impure metal is placed at the anode, and impurities dissolve into the electrolyte, leaving behind pure metal at the cathode.
Reason 2: The anode in electrolytic reduction does not play a significant role in the extraction of the metal; instead, it is involved in other reactions.
Assertion 3: Electrolytic reduction is suitable for extracting highly reactive metals like aluminum. Reason 3: In electrolytic reduction, the metal ions in the compound are reduced at the cathode to form pure metal.
Assertion 4: Electrolytic refining can be used to extract metals from their ores. Reason 4: In electrolytic refining, impurities are oxidized at the anode and dissolve into the electrolyte, leaving behind pure metal at the cathode.
Here are the answers to the assertion and reason-based questions: Question 1 Answer: A. Both Assertion 1 and Reason 1 are true, and Reason 1 explains Assertion 1.
For more detail study visit
class 10 Metals and Non-Metals
0 Comments
Leave a Reply. |
Blog SeaRCH Link
All
Join Us For Update |