|
Bihar Intermediate Time Table 2014 Date Sheet - Bihar Board 12th Exam Time Table/Date Sheet : 1. Bihar School Examination Board (BSEB) will declare Bihar Intermediate exam date at official website of Bihar Board. BSEB Senior Secondary Examination year 2014 will be conducted from month of March 2014. Dear students Bihar School Examination Board provide full time 12th class exam time table publish very soon at the main web portal of the BSEB. All the students of 12th class can check their exam time table at official website - www.biharboard.net .
2. Bihar Intermediate exam schedule 2014 is notified for class 12th annual exams. Bihar inter exam routine 2014 is scheduled for 18th February to 1st March 2014 tentatively. 3. The Bihar School Examination Board is established for holding and conducting an examination at the end of the Secondary School stage for prescribing course of studies for such examination and for carrying out such other objects and duties as may be considered necessary for the purpose as stated in the Act , Rules and Regulations of the Board. Students of both classes like Matric and Intermediate visit the official website for Bihar School Examination Board Exam schedule/ time table/ date sheet March 2014. 4. Bihar Intermediate board exam time table or Date sheet schedule 2014 subject wise is yet to be updated. we have kept our eyes at www.biharboard.net or http://www.biharboard.bih.nic.in to see any announcement about Bihar 12th exam dates 2014 we will update. 5. Bihar Board Intermediate and 10th class Syllabus 2013 - 2014 - http://www.biharboard.bih.nic.in/syllabus.htm 6. Bihar Board 10th class Model Question Papers - http://www.biharboard.bih.nic.in/ModelQSClassXI.htm 7. Bihar Board Intermediate Model Question Papers - http://www.biharboard.bih.nic.in/ModelQSClassXII.htm 8. Candidates if you want to received all latest updates Bihar via Facebook, then join us on Facebook page now.Candidates future reference bookmark this page.
6 Comments
SUMMATIVE ASSESSMENT – II, 2014 Class – X, SOCIAL SCIENCE SAMPLE PAPER
Time allowed: 3 hours Maximum Marks: 90
1 marks Questions 1. Which of the following was not a feature of the Conservatism in Europe after 1815? (a) Established traditional systems of state and society (b) Efficient Bureaucracy (c) Modern army (d) Democracy OR Which of these was not the challenge faced by the new Republic of Vietnam ? (a) Attack of the French armies to regain control. (b) Division of Vietnam. (c) Fall of the Bao Dai regime (d) Young generation’s emigration to Japan. 2. Which of the following is false about the Hapsberg Empire ? (a) It ruled over Austria – Hungary (b) It comprised of Italian and German speaking people (c) It shared a strong common identity (d) All the groups had no common allegiance to the emperor. OR/ Identify the blind poet from Vietnam who wrote, “I would rather face eternal darkness than see the faces of traitors.†(a) Ngyuyen Dinh Chieu (b) Phan Chu Trinch (c) Ngo Dinh Diem (d) Phan Boi Chau 3. What did the idea of Satyagrah emphasise ? (a) Traditional way of mass agitation. (b) Passive resistance. (c) Appeal to the conscience of the oppressor. (d) Use of physical force to fight the oppressor. 4.Which of the following is not true regarding the non-wearing of Khadi in cities. (a) Did not appeal to all. (b) Could not boycott mill cloth for too long. (c) Alternative Indian Institutions were fast to come up. (d) More expensive. 5. Which one of the following minerals is largely derived from placer deposits ? (a) Gold (b) Magnesium (c) Bromine (d) Salt 6. Where does Fertilizer Corporation of India has Fertilizer Industry ? (a) Hazira in Gujarat (b) Jaipur in Rajasthan (c) Aurangabad in Maharashtra (d) Patna in Bihar. 7. What was the aim of popular movement in Nepal held in April 2006 ? (a) To restore democracy (b) To protest against price rise. (c) To recall the elected representatives (d)To have good ties with India 8. Which of the following organizations pressurized the government in Bolivia to give up its control of municipal water supply ? (a) International Monetary Fund (b) World Health Organization (c) World Bank (d) Multi National Company 9. Which one of the following is a challenge to Political Parties ? (a) Parties put forward different policies and programmes. (b) Parties shape public opinion. (c) Parties do not seem to offer a meaningful choice. (d) Parties contest elections. 3 marks Questions 10. “The nation building process in Germany had demonstrated the dominance of the Prussian state power.†Analyse any three characteristic features of Prussia‟s leadership role in movement for German unification OR/ Explain any three features of scholars revolt in 1868 in Vietnam. 1868 11. What was the Khilafat Agitation ? Why did Gandhiji give support to this agitation ? 12. How can we conserve our mineral resources for future generation? Explains any three ways. 13. Explain the importance of automobile industry in India. 14. What is the importance of the IT sector for the Indian economy? Explain. 15. Describe any three features of a Pressure Groups. 16.Why is a democratic government considered less efficient ? Give any three reasons. 17. Suggest any three ways to accommodate social diversity for making life more harmonious 18. Explain the main challenge to democracy in India. 19. „Rapid improvement in technologies has been a major factor that has stimulated the globalization process.‟ Support the statement with any three suitable examples. 20. How can free and fair trade be achieved? Explain. 21. Explain the three tier quasi judicial machinery setup under COPRA. 5 marks Questions 22. Assess the role of Mahatma Gandhi in the National Movement with special reference to the methods adopted by him. 23. Describe the effects of US entry into war in Vietnam for the Vietnamese as well on the life within USA. 24. Explain the significance of Border Roads. 25. Explain the deposit and loan activities of banks. 26. Compare the terms of credit for small -landless agricultural workers and medium farmers in a village 27. What was Romanticism? How was it linked to Nationalism? 28. Cheap and affordable credit is crucial for the countries development. Highlight the role of loans in reference to India. 29. How far has India succeeded in overcoming, the challenge of expansion before its democracy? Evaluate. Map Question 5 mark 30. on the political map of India. Identifying these features place with the help of the following information and write their correct names on the lines marked in the map. (a) The place where the Non – Cooperation Movement was called off. (b) The place where the Cotton Mill Workers Satyagraha took place. (c) The place where Gandhiji started the satyagrahs in favour of Cotton Mill Workers. (d) The place where INC session of 1920 September was held. (e) The place where the Salt – satyagraha ended Links related to Class – X, SOCIAL SCIENCE SAMPLE PAPER Model Answer by the Candidates of Secondary School/Sr. Secondary School Certificate Exam.20138/12/2013
 Assessment of Speaking and Listening (ASL) in English for Students Speaking Sample for practice --Class IX Specifications for Speaking--Class XI Speaking Sample for Class XI Listening Specifications--Class IX Audio track Class IX : Track 1 | Track 2 | Track 3 | Audio scripts and Answer Key - Sample worksheets for Class IX Listening Test SAMPLE WORKSHEET FOR LISTENING TEST - Class IX SAMPLE WORKSHEET FOR LISTENING TEST - Class IX Audio Tracks : Class XI : Track 1 | Track 2 Audio scripts and Answer Key for Listening Worksheets Class XI SAMPLE WORKSHEET FOR LISTENING TEST - Class XI SAMPLE WORKSHEET FOR LISTENING TEST - Class XI Descriptors for Assessment of Speaking.zip (Videos) Speaking Videos A : File 1 | File 2 | File 3 | File 4 B : File 1 | File 2 | File 3 C : File 1 | File 2 | File 3 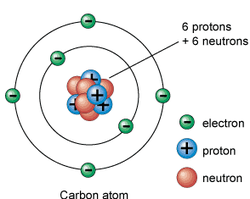 After completing this lesson, you will be able to:
The word atom originally meant a smallest possible particle of matter, not further divisible. Democritus 1. The existence of atoms was proposed as early as in the 5th century BC by the Greek philosophers Leucippus and his pupil Democritus, for which they were called atomists. 2. Democritus, develop the idea of atoms. He asked this question: If you break a piece of matter in half, and then break it in half again, how many breaks will you have to make before you can break it no further? . Democritus thought that it ended at some point, a smallest possible bit of matter. He called these basic matter particles, atoms. 4.The word "atom" is derived from the Greek word "atomos", which means indivisible". John Dalton 1. Five main points of Dalton's Atomic Theory • All matter is composed of extremely small particles called atoms. • All atoms of a given element are identical, having the same size, mass, and chemical properties. Atoms of a specific element are different from those of any other element. • Atoms cannot be created, divided into smaller particles, or destroyed. • Different atoms combine in simple whole-number ratios to form compounds. • In a chemical reaction, atoms are separated, combined, or rearranged. Weakness 1. Atoms consist of even smaller particles called electrons, protons and neutrons. 2. Atoms can be created and destroyed in the nuclear reactions such as nuclear fusion and nuclear fission. Atoms of the same element can have different physical properties, for example, isotopes of hydrogen. J.J. Thomson Plum pudding model 1. In physics, the Plum pudding model of the atom was made after the discovery of the electron and was proposed by the discoverer of the electron, J. J. Thomson. 2. In it, the atom is envisioned as electrons surrounded by a soup of positive charge, like plums surrounded by pudding. 3. The electrons were positioned uniformly throughout the atom. 4. Instead of a soup, the model is also said to have had a cloud of positive charge. 5. This model can be compared to a British treat called plum pudding, hence the name. It is also known as the chocolate chip cookie model. Ernest Rutherford Gold foil experiment The Gold foil experiment, or Geiger-Marsden experiment was an experiment done by Hans Geiger and Ernest Marsden in 1909, under the direction of Ernest Rutherford at the Physical Laboratories of the University of Manchester which led to the downfall of the plum pudding modelof the atom. 1. They measured the deflection of alpha particles directed normally onto a sheet of very thin gold foil. 2. Under the prevailing plum pudding model, the alpha particles should all have been deflected by at most of a few degrees. 3. However they observed that a very small percentage of particles were deflected through angles much larger than 90 degrees. 4. From this Rutherford concluded that the atom contained a very small positive charge which could repel the alpha particles if they came close enough. Rutherford Atom 1. Early in 1911 Rutherford published a revised model of the atom, known as the Rutherford atom. 2. He concluded that • the atom is mostly empty space, • most of the atom's mass concentrated in a tiny center, the nucleus and electrons being held in orbit around it by electrostatic attraction. • The nucleus was around 10-15 meters in diameter, in the centre of a 10-10 metre diameter atom. • Those alpha particles that had come into close proximity with the nucleus had been strongly Model: Solar System Model (Bohr’s Model) 1. Niels Bohr improved on Rutherford's atomic model. 2. Bohr model depicts the atom as a small, positively charged nucleus surrounded by electrons in orbit - similar in structure to the solar system, but with electrostatic forces providing attraction, rather than gravity. 3. According to Bohr’s Model • Electrons in an atom of an element are not randomly distributed around the atomic nucleus. • Electrons move around the nucleus in fixed orbits.• Each orbit forms a circle and has a fixed distance from the nucleus. James Chadwick’s Model In 1932, James Chadwick bombarded beryllium (Be) with alpha aprticles. He allowed the radiation emitted by beryllium to incident on a paraffin wax. It was found that protons were shot out form the paraffin wax. People began to look for what was in the "beryllium radiations". 1. Chadwick discovered the presence of neutrons in the nucleus. 2. He concluded that the nucleus contains another tiny particle known as a neutron that has nocharge. 3. The neutron mass is almost similar to the proton mass. 4. All nuclei contain protons and neutrons, except for the hydrogen which contains protons. only Modern Atomic Model 1. The atomic model in the present day is based on the contributions of the above scientists. 2. According to the modern atomic model, (a) The central nucleus consists of protons and neutrons. It containing almost all the mass of the atom. (b) the nucleus of an atom is very small compared to the size of the atom (c) the electrons are orbiting outside the nucleus in the electron shells(d) the electrons are moving in electron shells at a very high speed and we cannot determine the position of the electrons at a particular time The subatomic particles of an atom 1. Atoms are made up of tiny particles called subatomic particles. 2. An atom contains three types of subatomic particles:a. proton,b. neutron andc. electron, 3. The proton and neutron form the nucleus at the centre of an atom. 4. The electron moves around the nucleus at a very high speed. 5. The nucleus is positively charged because of the presence of protons, which are positively charged. The neutrons are neutral. Proton number and nucleon number Proton Number 1. The proton number (Z) represent the number of protons found in the nucleus of an atom. Proton number = the number of protons 2. The proton number is also known as the atomic number. 3. In an atom of neutral charge, the number of electrons also equals the atomic number. 4. Hence, the proton number of an atom can also represent the number of electrons. Nucleon Number 1. The nucleon number (A), also called atomic mass number or mass number, is the number ofprotons plus the number of neutrons in an atomic nucleus. Nucleon number = Number of protons + Number of Neutrons The nucleon number of an atom is about the same as the mass of the atom because the mass of anelectron is very small and can be ignored. Continued>>>>>>>>>> Read more to excel IX Atomic Structure ( a fundamental particles) Discovery of Electron 9th Atomic Structure (Discovery of a fundamental particle protons) 9th Atoms and Molecules 9th Structure of Atoms Practice paper and sample paper based On Atomic structure Discovery of electrons, protons and neutrons Download File
|
Blog SeaRCH Link
All
Join Us For Update |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||






