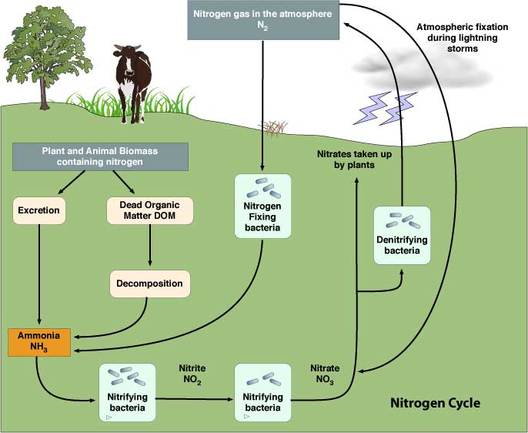
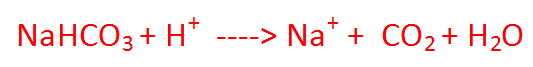 Source: sciencebitz.com Source: sciencebitz.com Nitrogen Fixation: Nitrogen Fixation is the conversion of atmospheric nitrogen (N2) into reactive compounds such as ammonia (NH3) and nitrate (NO3-). The breaking of the bonds between the nitrogen atoms requires a great deal of energy and occurs naturally in two primary ways: 1. Abiotic Fixation: Nitrate is the result of high energy fixation in the atmosphere from lightning and cosmic radiation. In this process, N2 is combined with oxygen to form nitrogen oxides such as NO and NO2, which are carried to the earth’s surface in rainfall as nitric acid (HNO3). This high energy fixation accounts for approximately 10% of the nitrate entering the nitrogen cycle. 2. Biological fixation: Biological fixation is accomplished by a series of soil micro-organisms such as aerobic and anaerobic bacteria. Often, symbiotic bacteria such as Rhizobium are found in the roots of legumes and provide a direct source of ammonia to the plants. In root nodules of these legumes, the bacteria split molecular nitrogen into two free nitrogen atoms, which combine with hydrogen to form ammonia (NH3). The following plants are common examples of legumes: clover, alfalfa, soy beans, and chick peas. The breakdown of these legumes by bacteria during ammonification actually returns excess nitrogen not utilized by the plant to the surrounding soil. Therefore, to promote sustainable soil fertility, it is beneficial to use these agricultural crops in rotation with other plants, such as corn, that are more profitable but deplete the available nitrogen in the soil-Some free-living aerobic bacteria, such as Azotobacter, and anaerobic bacteria, like Clostridium, freely fix nitrogen in the soil and in aquatic environments. Some members of the photosynthetic Cyanobacteria phylum fix nitrogen in aquatic environments as well. Nitrification: Nitrification is the process by which ammonia is oxidized to nitrite ions (NO2-) and then to nitrate ions (NO3-), which is the form most usable by plants. The two groups of micro-organisms involved in the process are Nitrosomas and Nitrobacter. Nitrosomas oxidize ammonia to nitrite and Nitrobacter oxidize nitrite to nitrate. Assimilation: Nitrates are the form of nitrogen most commonly assimilated by plants through root hairs. Since heterotrophic organisms cannot readily absorb nitrogen as plants do, they rely on acquiring nitrogen-based compounds through the food they eat. Since plants are the base of the food chain, the nitrogen-based compounds they have assimilated into their tissue will continue to pass from one organism to another (through consumption) as matter and energy transfers through the ecosystem’s food web. Ammonification: In ammonification, a host of decomposing microorganisms, such as bacteria and fungi, break down nitrogenous wastes and organic matter found in animal waste and dead plants and animals and convert it to inorganic ammonia (NH3) for absorption by plants as ammonium ions. Therefore, decomposition rates affect the level of nutrients available to primary producers. Denitrification: Denitrification is the process by which nitrates are reduced to gaseous nitrogen (N2) and lost to the atmosphere. This process occurs by facultative anaerobes in anaerobic environments. Farmers with waterlogged fields and soils that have high clay content are especially vulnerable to nitrogen losses due to denitrification.
0 Comments
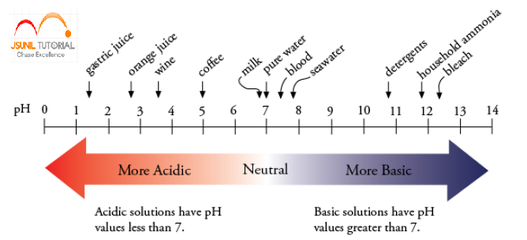 Question: The pH of hydrochloric acid solution is 3. Does it mean that it has only hydronium ions? If not, how are OH- ions generated? Answer: It is found that for any aqueous solution (neutral, acidic or basic), the product of H+ ion concentration and OH- ion concentration is always constant and equal to 10-14 at 25°C.This is called ionic product of water(Kw) Kw= [H+] [OH - ] = 10-14 Here The pH of hydrochloric acid solution is 3. Let OH- ion concentration = p [H+] [OH - ] = 10-14 1.0 × 10-3 x p = 10-14 p = [10-14]¸[ 1.0 × 10-3] =1.0 × 10-11 Thus for a solution if H+ concentration is known, OH- ion concentration is fixed (because their product is always constant).Hence we express the acidic, basic or neutral character of any solution by expressing its H+ ion concentration in terms of pH where p stands for potenz ie power.  Question: What is aqua-regia ? Answer: A mixture of concentrated nitric and hydrochloric acids. It is a highly corrosive liquid able to attack gold and other resistant substances. Question: Plaster of Paris should be stored in a moisture-proof container. Explain why? Answer: Plaster of paris should be stored in moisture proof container because Plaster of paris is powdered form of gypsum with less water of crystallization. But when we keep in open it will react with water to for hard solid mass Gypsum. CaSO4. 1/2H2O + 3/ H2O ------- > CaSO4. 2H2O Question: Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. Amount and concentration taken for both the acids are same. In which test tube will the fizzing occur more vigorously and why? Answer:-In test tube A fizzing occur more vigorously. Because HCl is stronger acid than acetic acid. Hence, HCl liberates hydrogen gas more vigorously, which causes fizzing more vigorously. What will happen if heating is not controlled while preparing Plaster of Paris from gypsum?19/6/2014  Baking soda is a single compound which has sodium hydrogen carbonate which is alkaline (basic) in nature . Baking powder is a mixture of sodium hydrogencarbonate and a mild edible(eatable) acid such as tartaric acid. When baking powder mixes with water, then the sodium hydrogencarbonate reacts with tartaric acid to evolve carbon dioxide gas which gets trapped in the wet dough and bubbles out slowly making the cake to rise and hence 'soft and spongy'.The equation which takes place can be shown as: |