 After completing this lesson, you will be able to:
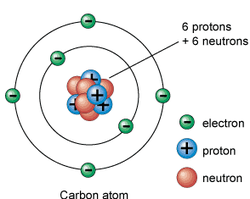
The word atom originally meant a smallest possible particle of matter, not further divisible. Democritus 1. The existence of atoms was proposed as early as in the 5th century BC by the Greek philosophers Leucippus and his pupil Democritus, for which they were called atomists. 2. Democritus, develop the idea of atoms. He asked this question: If you break a piece of matter in half, and then break it in half again, how many breaks will you have to make before you can break it no further? . Democritus thought that it ended at some point, a smallest possible bit of matter. He called these basic matter particles, atoms. 4.The word "atom" is derived from the Greek word "atomos", which means indivisible". John Dalton 1. Five main points of Dalton's Atomic Theory • All matter is composed of extremely small particles called atoms. • All atoms of a given element are identical, having the same size, mass, and chemical properties. Atoms of a specific element are different from those of any other element. • Atoms cannot be created, divided into smaller particles, or destroyed. • Different atoms combine in simple whole-number ratios to form compounds. • In a chemical reaction, atoms are separated, combined, or rearranged. Weakness 1. Atoms consist of even smaller particles called electrons, protons and neutrons. 2. Atoms can be created and destroyed in the nuclear reactions such as nuclear fusion and nuclear fission. Atoms of the same element can have different physical properties, for example, isotopes of hydrogen. J.J. Thomson Plum pudding model 1. In physics, the Plum pudding model of the atom was made after the discovery of the electron and was proposed by the discoverer of the electron, J. J. Thomson. 2. In it, the atom is envisioned as electrons surrounded by a soup of positive charge, like plums surrounded by pudding. 3. The electrons were positioned uniformly throughout the atom. 4. Instead of a soup, the model is also said to have had a cloud of positive charge. 5. This model can be compared to a British treat called plum pudding, hence the name. It is also known as the chocolate chip cookie model. Ernest Rutherford Gold foil experiment The Gold foil experiment, or Geiger-Marsden experiment was an experiment done by Hans Geiger and Ernest Marsden in 1909, under the direction of Ernest Rutherford at the Physical Laboratories of the University of Manchester which led to the downfall of the plum pudding modelof the atom. 1. They measured the deflection of alpha particles directed normally onto a sheet of very thin gold foil. 2. Under the prevailing plum pudding model, the alpha particles should all have been deflected by at most of a few degrees. 3. However they observed that a very small percentage of particles were deflected through angles much larger than 90 degrees. 4. From this Rutherford concluded that the atom contained a very small positive charge which could repel the alpha particles if they came close enough. Rutherford Atom 1. Early in 1911 Rutherford published a revised model of the atom, known as the Rutherford atom. 2. He concluded that • the atom is mostly empty space, • most of the atom's mass concentrated in a tiny center, the nucleus and electrons being held in orbit around it by electrostatic attraction. • The nucleus was around 10-15 meters in diameter, in the centre of a 10-10 metre diameter atom. • Those alpha particles that had come into close proximity with the nucleus had been strongly Model: Solar System Model (Bohr’s Model) 1. Niels Bohr improved on Rutherford's atomic model. 2. Bohr model depicts the atom as a small, positively charged nucleus surrounded by electrons in orbit - similar in structure to the solar system, but with electrostatic forces providing attraction, rather than gravity. 3. According to Bohr’s Model • Electrons in an atom of an element are not randomly distributed around the atomic nucleus. • Electrons move around the nucleus in fixed orbits.• Each orbit forms a circle and has a fixed distance from the nucleus. James Chadwick’s Model In 1932, James Chadwick bombarded beryllium (Be) with alpha aprticles. He allowed the radiation emitted by beryllium to incident on a paraffin wax. It was found that protons were shot out form the paraffin wax. People began to look for what was in the "beryllium radiations". 1. Chadwick discovered the presence of neutrons in the nucleus. 2. He concluded that the nucleus contains another tiny particle known as a neutron that has nocharge. 3. The neutron mass is almost similar to the proton mass. 4. All nuclei contain protons and neutrons, except for the hydrogen which contains protons. only Modern Atomic Model 1. The atomic model in the present day is based on the contributions of the above scientists. 2. According to the modern atomic model, (a) The central nucleus consists of protons and neutrons. It containing almost all the mass of the atom. (b) the nucleus of an atom is very small compared to the size of the atom (c) the electrons are orbiting outside the nucleus in the electron shells(d) the electrons are moving in electron shells at a very high speed and we cannot determine the position of the electrons at a particular time The subatomic particles of an atom 1. Atoms are made up of tiny particles called subatomic particles. 2. An atom contains three types of subatomic particles:a. proton,b. neutron andc. electron, 3. The proton and neutron form the nucleus at the centre of an atom. 4. The electron moves around the nucleus at a very high speed. 5. The nucleus is positively charged because of the presence of protons, which are positively charged. The neutrons are neutral. Proton number and nucleon number Proton Number 1. The proton number (Z) represent the number of protons found in the nucleus of an atom. Proton number = the number of protons 2. The proton number is also known as the atomic number. 3. In an atom of neutral charge, the number of electrons also equals the atomic number. 4. Hence, the proton number of an atom can also represent the number of electrons. Nucleon Number 1. The nucleon number (A), also called atomic mass number or mass number, is the number ofprotons plus the number of neutrons in an atomic nucleus. Nucleon number = Number of protons + Number of Neutrons The nucleon number of an atom is about the same as the mass of the atom because the mass of anelectron is very small and can be ignored. Continued>>>>>>>>>> Read more to excel IX Atomic Structure ( a fundamental particles) Discovery of Electron 9th Atomic Structure (Discovery of a fundamental particle protons) 9th Atoms and Molecules 9th Structure of Atoms Practice paper and sample paper based On Atomic structure Discovery of electrons, protons and neutrons Download File
1 Comment
|
Blog SeaRCH Link
All
Join Us For Update |
||||||


