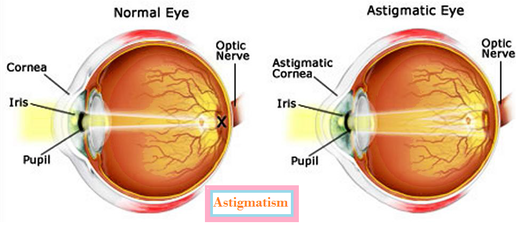
 Q. The near point of a hypermetropic eye is 1 m. What is the power of the lens required to correct this defect? Assume that the near point of the normal eye is 25 cm. Answer: In the problem it is given that the near point of the normal eye is 25 cm, hence u = - 25 cm. The near point of a hypermetropic eye is 1 m . It is the nearest point up to which the hyper me tropic eyes can see object. So to correct hypermetropic eyes of this defect, person has to use Spectacles with convex lens of suitable focal length. This lens form virtual image of nearby object at the near point of hypermetropic eye. so image distance is 1m=100cm v= near point of hypermetropic eye = -1m = -100 cm u = near point of normal eye = -25cm. 1/f = 1/v -1/u = -1/100 + 1/25 = (-1 + 4)/100 =3/100 f = 100/3 cm = 1/3 m Power = 1/f = 1/(1/3) =3 dioptre Q. The far point of a myopic person is 80 cm in front of the eye. What is the nature and power of the lens required to correct the problem? Answer: This defect is corrected by concave lens. For myopic personthe object distance is ¥ since the far point this eyes is 80 cm. So to correct myopic eyes of this defect, person has to use Spectacles with concave lens of suitable focal length. This lens form virtual image of distant object at the far point of myopic eye. So image distance is 80 cm so applying lens formula 1/v - 1/u = 1/f v = - 80 cm u = infinity=0 -1/80 + 1/o = 1/f 1/f = -1/80 f = -80 cm or -0.8 m power is = -1/0.8 = -10/8 = -1.25 D So it would be a concave lens as it has a negative power Why is the colour of the clear Sky Blue? The blue light has lest velocity. Thus, when sunlight passes through the atmosphere, the fine particles in air scatter the blue colour (shorter wavelengths) more strongly. Hence the colour of the clear Sky appears blue. If there is no atmosphere, there would not have been any scattering. Then, the sky would have looked dark. Explain reason for the reddish appearance of the Sun at the sunrise or sunset. At the sunrise or the sunset, Light from the Sun has to cover larger distance in the earth’s atmosphere before reaching our eyes. Since red colour light has longer wave length deviate lesser then other colours light and reach to our eyes. What is Persistence of vision? Ans: The impression of image is remains on retina for about 1/16th of second. This is used during cinematography. The sequence of still picture taken by movie camera is projected on the screen at he rate of 24 or more images per second so that images merge in one another on screen . What is astigmatism? In astigmatism a person can see only in one plane either horizontal or vertical. When light rays enter the eye they do not focus correctly on the retina, resulting in a blurred image. Astigmatism may also be caused by an irregularly shaped lens, which is located behind the cornea. 10th chapter wise solution
2 Comments
Guess Paper – 2012 Class – X Subject –Social Science
Answer the following questions:- 1. When was the Champaran Movement started by Mahatma Gandhi and why? 1mark 2. Why was policy of non-cooperation adopted by Gandhiji and Congress? 1-mark 3. Why did the Indians oppose the Simon Commission? 1-mark 4. When was a satyagraha organized in the kheda district of Gujrat and why? 1-mark 5. Why did the industrial workers not join Civil Disobedience Movement? 1-mark 6. How did people belonging to different communities, regions or language groups develop a sense of collective belonging in India? 3-marks 7. Who formed the Swaraj Party and why? 3-marks 8. Describe the policy of the government during the first phase of the Civil Disobedience Movement. 3-marks 9. Describe the views of Mahatma Gandhi on untouchability. 3-marks 10. Describe the events leading to the Salt March and the Civil Disobedience Movement in 1930 3-marks 11. Why Gandhiji decided to withdraw the Non-Cooperation Movement? 4-marks 1. What do you understand by modern nationalism in Europe? 1-mark 2. Name the famous brothers who were Muslim leaders in 1919. 1-mark 3. When and why a satyagraha movement was organized in Ahmedabad? 1-mark 4. Who was the spiritual head of the Islamic world? 1-mark 5. Describe Gandhiji’s view on the role of women in life. 1-mark 6. Name two prominent leaders who formed the Swaraj Party within the Congress. What was the aim of the Swaraj Party? 3-marks 7. What do you understand by boycott? Why was foreign cloth boycotted during Non-Cooperation Movement? 3-marks 8. Describe Mahatma Gandhi’s view on satyagraha. 3-marks 9. Describe the events leading to Non-Cooperation Movement in support of Khilafat as well as for Swaraj 3-marks 10. Write a short note on Simon Commission. 3-marks 11. How the first World War helped in the growth of the National Movement in India?4-marks Class – X Subject –History (a) “ A large city population was thus both a threat and an opportunity.” Justify this statement. (b) What led to the major expansion of Bombay’s population in the mid-18th century ? (c) Who wrote “The Bitter Cry of Outcast London’ and when ? d) What do you know about ‘London Season’ ? e) Explain in brief the history of Land Reclamation in Mumbai. (f) Explain the social change that took place in the 19th century which resulted into political movements. (g) When and where was the very first section of the underground railway in the world opened ? Why did people oppose it ? (h) What forms of entertainment came up in England came up in England to provide leisure activities for the people ? (i) “Many felt that the ‘Iron Monsters’ added to the mess and unhealthiness of the city.” Explain. (j) Who developed the concept of the principle of the Garden city of New Earswick ? Mention its features. (k) Who did the first social survey of low skilled London workers in the East End of London ? Explain his suggestions in the context of what he surveyed about. (l) ‘London grew as a crime flourished city’. Discuss the statement. “ Bombay films have contributed in a big way to produce an image of the city as a blend of dream and reality of slums and star bunglows.” Explain. social science_sample_paper x-1 social_science_sample_paper_x-2 social_science_sample_paper_x-3 social_science_sample_paper_x-4 social_science_sample_paper_x-5 social_science_sample_paper_x-6 social_science_sample_paper_x-7 social_science_Study material and N0tes  CLASS X SCIENCE (Physics+Chemistry) SUMMATIVE ASSESSMENT TERM II 2012 SAMPLE PAPER- I 1. Why carbon forms strong bonds with other carbon atoms, hydrogen, oxygen, nitrogen or sulphur? 2. Name the part of eye responsible for conversion of light into electrical impulses. 3. a. How does valency vary in a group on going from top to bottom? b. How does atomic size vary in a period on going from left to right? 4. Write one property of hydrogen which makes it resemble with (a) Alkali metals (b) Halogens 5. a Arrange the following common substances in the increasing order of refractive indices. Ice, Kerosene, Glass, Diamond, Alcohol, Waterb. Is it necessary that optically dense medium possesses greater mass density? Give an example. 6. On reaction with sodium hydroxide, X yielded Ethanoic acid and ethanol.(a) Give the IUPAC name of X? (b) Name the reaction. (c) Give a chemical reaction for the above reaction. 7. (a) How does the electronic configuration of an atom related to its position in the Modern periodic table? Give one example. (b) Why nitrogen is more electronegative than phosphorus? 8. A concave length has focal length of 25 cm. At what distance should the object from the lens be placed so that if it forms an image at 20 cm distance from the lens? Also find the magnification produced by the lens. 9. Give an explanation for the formation of a rainbow. 10. How are we able to see distant and near by objects clearly? Which part of eye helps in changing curvature of lens? Why no image is formed at blind spot? 11.(a) Why magnification is taken negative for real images and positive for virtual images? (2) (b) Why convex mirror is used in rear view mirrors and not concave mirror? (2)(c) Power of concave lens is 4.5 D. Find its focal length. (1) 12.(a) Find the size, nature and position of image formed when an object of 1 cm is placed at a distance of 15 cm from a concave mirror of focal length 10 cm. (3) (b) Why does light travels faster in water in comparison to kerosene. (Refractive index of water and kerosene are 1.33 and 1.44 respectively) (2) 13 . a. Which property of carbon leads to formation of large number of compounds? Define itb. What is the functional group in the following molecules? i. CH3CH2CH2OH ii. CH3COOH c. Which of the following formula represents a saturated hydrocarbon? CnH 2n, C nH2n+1 Cn H2n+2 Cn H2n-2d. What happens when methane is burnt in oxygen?e. Why is the conversion of ethanol to Ethanoic acid an oxidation reaction? 14. a. Give three points to distinguish between alkenes and alkynes. b. Explain the mechanism of cleaning action of detergents CLASS X SCIENCE (Physics & Chemistry) SUMMATIVE ASSESSMENT TERM II 2012 SAMPLE PAPER- I (solution) 1. Due to small size and presence of four valence electrons, carbon forms strong bonds with other carbon atoms, hydrogen, oxygen, nitrogen or sulphur. 2. Retina. 3. a. Valency remains same on moving from top to bottom in a particular group. This is because the outermost electronic configuration of all the elements in a group remains same. b. Atomic size decreases on moving from left to right in a period. This is because the number of shells remains same but the nuclear charge increases. Due to this, electrons are pulled closer to the nucleus and decrease the atomic size. 4. Resemblance with alkali metals: Hydrogen has the same outermost electronic configuration as that of alkali metals.Resemblance with halogens:Hydrogen exists as diatomic molecule as halogens. 5. a. In the order of increasing refractive indices:Ice; Water; Alcohol; Kerosene; Glass; Diamond b. No, it is not always necessary. For e.g., kerosene and turpentine oil having high refractive index are optically dense than water. But its mass density is less than that of water. That is why oil floats on the surface of water. 6. (a) X is ethyl Ethanoate. (b) It is Saponification reaction. (c) CH3COOC2H5 + Na OH -----------> C2H5OH + CH3COOH 17. (a) By the electronic configuration of an atom we can identify the group and period to which the atom belongs. For example - Oxygen has atomic number 8. Its electronic configuration is 2,6. As it can gain two electrons to complete its octet, so it belongs to group 16 Also, since the electrons are filled in two shells. So. Oxygen belongs to the second period. (b) As we move down the group, electro negativity of elements decreases as the atomic size increases. Nitrogen is placed above phosphorus in group 15. So, nitrogen is more electronegative than phosphorus. 8. A concave lens always forms a virtual, erect image on the same side of the object.v = -20 cm, f = -25 cm, u = ? 1/v -1/u = 1/f 1/u = 1/(-20) – 1/(-25) 1/u = -1/100 u= -100cm Thus object distance is 100 cm. Magnification= v/u = -20/ (-100) = + 0.5 1 Thus image is erect, virtual and is half of the size of object. 9. Formation of rainbow: The rainbow is formed in the sky when sun shines and it is raining at the same time. The raindrops in the atmosphere act like many small prisms. As white light enters and leaves these raindrops the various colours present in white light are refracted by different amount due to which an arch of seven colours called rainbow is formed in the sky. Diagram 10. (a) Accommodation (b) Ciliary muscles. (c) No image is formed at the blind spot because no nerve cells are present there to carry the information of image to the brain. 11. (a) Distance measured upward and perpendicular to the principal axis are taken as positive. Distances measured down ward and perpendicular to the principal axis are taken as negative. So magnification for a real image is taken negative and for a virtual image it is taken as positive. (b) A convex lens forms a virtual and small sized image of the object and concave mirror forms real and inverted image. Thus by using convex mirror we can get view of wider field which is not possible in case of a concave mirror. (c) Power = 1/Focal length Or Focal length = 1/Power f= 1/4.5 f = 0.22 cm 12. (a) Object distance, u = -15 cm Image distance, v= ? Focal length, f = -10 cm Mirror formula, 1/v + 1/u = 1/f 1/v + 1/ (-15) = 1/(-10) 1/v = -1/10 + -/15 1/v = -3+2/30 1/v = -(1/30) Thus the position of image is formed on left hand side in front of the concave mirror at a distance of30 cm, its nature will be real and inverted. Size of image m= -v/u m= - [(-30)/(-15)] m= -2.Thus the size of image is 2 cm and image is real and inverted. (b) Refractive index of a medium= Speed of light in air/ Speed of light in medium.So, speed of light in medium= speed of light in air/ refractive index Thus speed of light will be maximum in medium having lowest refractive index. Therefore speed of light is more in water in comparison to kerosene. 13. (a) Catenation The property of carbon atom to link with other carbon atoms to form large molecules is called as catenation. b. i. Functional group is –OH group. ii. Functional group is –COOH group. C. Cn H2n+ 2 represents a saturated hydrocarbon. d. Methane burns in oxygen with formation of carbon dioxide and water. CH4 + 2O2 -----------> CO2 + 2H2O e. Conversion of ethanol to Ethanoic acid is an oxidation reaction because oxygen is added to ethanol and Ethanoic acid is formed. CH 3CH2 OH + O2 -----------> CH3 COOH + H2 O Ethanol Ethanoic acid 14. a. No. Alkene (1) Unsaturated hydrocarbon containing a double bond between two carbon atoms are known as alkenes. (2) General formula is CnH2n Example: Ethene, Propene Alkynes: Unsaturated hydrocarbons containing a triple bond between two carbon atoms are known as alkynes. (2) General formula is CnH2n -2 (3) Example: Ethyne, Propyne b. A detergent molecule consists of two ends – (a) hydrocarbon tail which is hydrophobic (water repelling) and (b) polar head which is hydrophilic (water attracting or loving). When a detergent is dissolved in water, the hydrocarbon tail aligns itself towards the dirt and ionic part aligns itself towards the water. The molecules gather together as clusters, called micelles. When water is agitated, the dirt suspended in the micelles is easily rinsed away. Thus, the cloth gets cleaned. CBSE BOARD EXAM SPECIAL Class X: Sample Question Papers for Term I (September ) Class X: Sample Question Papers for Term I (September ) Class IX: Sample Question Papers for Term I (September ) Class IX: Sample Question Papers for Term I (September ) Class X: Sample Question Papers for Term II (March ) Class X: Sample Question Papers for Term II (March ) Class IX: Sample Question Papers for Term II (March ) Class IX: Sample Question Papers for Term II (March ) Class IX-X: Value Based Questions PERIODIC PROPERTIES
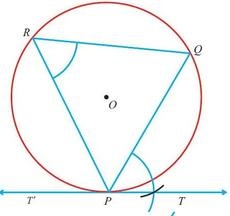
in a period the number of valence electrons and the nuclear charge increases from left to right. It increases the force of attraction between them. In a group the number of filled shells increases and valence electrons are present in higher shells. This decreases the force of attraction between them and the nucleus of the atom. These changes affect various properties of elements and they show gradual variation in a group and in a period and they repeat themselves after a certain interval of atomic number. Such properties are called periodic properties. Atomic radius What happens to atomic radii in a group and period and why? Atomic radius is the distance between the centre of atom and the outermost shell. In a period, atomic radius generally decreases from left to right. 2nd Period Li Be B C N O F 155 112 98 91 92 73 72 3rd Period Na Mg Al Si P S Cl 190 160 143 132 128 127 99 In a period there is a gradual increase in the nuclear charge. Since valence electrons are added in the same shell, they are more and more strongly attracted towards nucleus. This gradually decreases atomic radii. Atomic radii increase in a group from top to bottom. Element Atomic radius Element Atomic radius Li 155 F 72 Na 190 Cl 99 K 235 Br 114 Rb 248 I 133 As we go down a group the number of shells increases and valence electrons are present in higher shell and the distance of valence electrons from nucleus increases. Also, the number of filled shells between valence electrons and nucleus increases Both the factors decrease the force of attraction between nucleus and valence electron. Therefore, atomic size increases on moving down a group. Ionization energy Negatively charged electrons in an atom are attracted by the positively charged nucleus. For removing an electron this attractive force must be overcome by spending some energy. The minimum amount of energy required to remove an electron from a gaseous atom in its ground state to form a gaseous ion is called ionization energy. It is measured in unit of kJ mol-1. It is a measure of the force of attraction between the nucleus and the outermost electron. Stronger the force of attraction, greater is the value of ionization energy. It corresponds to the following process: If only one electron is removed, the ionization energy is known as the first ionization energy. If second electron is removed the ionization energy is called the second ionization energy. Ionization energy decreases in a group from top to bottom This is because the force of attraction between valence electrons and nucleus decreases in a group from top to bottom. The ionization energy increases in a period from left to right. This is because the force of attraction between valence electron and nucleus increases in a period from left to right. Electron affinity: The tendency of element to gain an additional electron. This ability is measured by electron affinity. It is the energy change when an electron is accepted by an atom in the gaseous state. Electron affinity is assigned a positive value when energy is released during the process. Greater the value of electron affinity, more energy is released during the process and greater is the tendency of the atom to gain electron. In a group, the electron affinity decreases on moving from top to bottom as less and less amount of energy is released. In a period, the electron affinity increases from left to right, as more and more amount of energy is released Electro negativity Electro negativity is relative tendency of a bonded atom to attract the bond-electrons towards it self. It just compares the tendency of various elements to attract the bond-electrons towards themselves. Electro negativity decreases in a group from top to bottom. Electro negativity increases in a period from left to right. Electro negativity is related to ionization energy. Electrons with low ionization energies have low electronegativity because their nuclei do not exert a strong attractive force on electrons. Elements with high ionization energies have high electronegativity due to the strong pull exerted on electrons by the nucleus. Oxides and its nature: Metals react with oxygen to form oxides by loss of electrons. These oxides on dissolution in water form bases. Reactivity of elements: Down the group reactivity of metals increases as the tendency to lose electrons increases due to increased atomic size. Reactivity of non- metals decreases down the group because of the increased atomic size and the tendency to gain electrons decreases. On moving across the period, the reactivity first increases due to the decrease in the metallic character and increase in non metallic character. Metallic and Non-metallic Properties The metallic character of an element is expressed in terms of its electron releasing tendency while non-metallic character in term of electron accepting tendency. In group metallic character of the element increases down the group due to increasing atomic size or because outermost electrons are farther away from the nucleus .So they can be easily lost. In periods metallic character of the element decreases along a period due to decrease in atomic size along a period or outermost electrons are closer to nucleus. So they can not be easily lost. Periodic Properties of the Elements –X Chemistry by JSUNIL Periodic Properties Check your understanding CBSE X CONSTRUCTIONS (Maths) Division of a Line Segment ,Construction of Tangents to a Circle,To construct a triangle similar to a given triangle as per given scale factor,To construct the tangents to a circle from a point outside it. You will learn to do the following constructions : 1. To divide a line segment in a given ratio. 2. To construct a triangle similar to a given triangle as per given scale factor which may be less than or may be greater than 1. 3. To construct tangent at a point on a given circle. 4. To construct the pair of tangents from an external point to a circle Q. Draw a circle of radius 3.2 cm. Take a point P on this circle and draw a tangent at P.(using the centre) Given: Radius of the circle = 3.2 cm.
Steps of construction(i) With O as the centre draw a circle of radius 3.2 cm. (ii) Take a point P on the circle and join OP. (iii) Draw an arc of a circle with centre at P cutting OP at L. (iv) Mark M and N on the arc such that m(arc L M ) = m(arcMN) (v) Draw the bisector PT of the angle <MPN (vi) Produce TP to Tlto get the required tangent T'PT. 10th Geometry topic -Construction [Guess Question paper for CBSE Board] Download File |
Blog SeaRCH Link
All
Join Us For Update |